Difference between revisions of "SpoIVA"
| Line 129: | Line 129: | ||
* '''Additional information:''' | * '''Additional information:''' | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 276 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 10 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 309 {{PubMed|21395229}} | ||
=Biological materials = | =Biological materials = | ||
| − | |||
* '''Mutant:''' | * '''Mutant:''' | ||
Revision as of 14:14, 17 April 2014
- Description: ATPase, spore coat morphogenetic protein, anchors the spore coat to the spore surface via SpoVM
| Gene name | spoIVA |
| Synonyms | spoVP |
| Essential | no |
| Product | ATPase, basement layer protein for spore coat assembly |
| Function | spore cortex formation and coat assembly |
| Gene expression levels in SubtiExpress: spoIVA | |
| Interactions involving this protein in SubtInteract: SpoIVA | |
| MW, pI | 55 kDa, 4.546 |
| Gene length, protein length | 1476 bp, 492 aa |
| Immediate neighbours | hbs, yphF |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 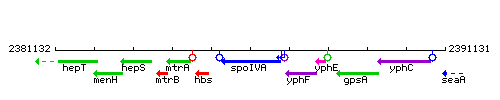
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed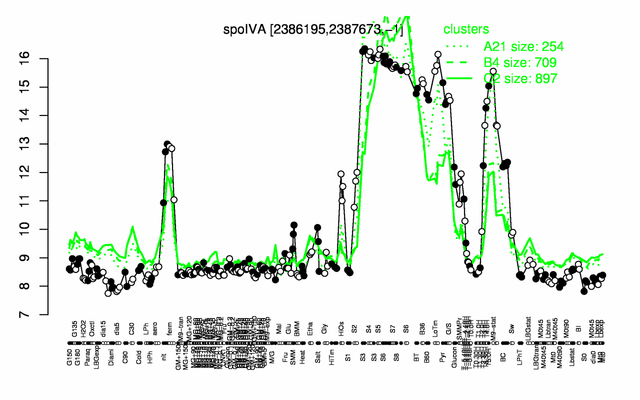
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
SpoIVA-dependent proteins of the spore coat basement
The gene
Basic information
- Locus tag: BSU22800
Phenotypes of a mutant
- the spore coat does not localize to the spore surface but self-assembles into aggregates in the mother cell cytoplasm PubMed
Database entries
- BsubCyc: BSU22800
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- uses ATP hydrolysis to drive self-assembly into static filaments PubMed
- ATP hydrolysis drives polymerization of a nucleotide-free filament PubMed
- ploymerization depends on a critical threshold concentration of SpoIVA that is only achieved once the protein is recruited to the surface of the developing spore PubMed
- Protein family:
- belongs to the TRAFAC class of P-loop GTPases, but has lost the ability to bind GTP PubMed
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains: contains a Walker A ATPase domain
- Modification:
- Effectors of protein activity:
- Localization:
- innermost protein of the spore coat basement PubMed
Database entries
- BsubCyc: BSU22800
- Structure:
- UniProt: P35149
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: spoIVA PubMed
- Additional information:
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 276 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 10 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 309 PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications