LeuB
Revision as of 14:09, 17 April 2014 by 134.76.70.252 (talk)
- Description: 3-isopropylmalate dehydrogenase
| Gene name | leuB |
| Synonyms | leuC |
| Essential | no |
| Product | 3-isopropylmalate dehydrogenase |
| Function | biosynthesis of leucine |
| Gene expression levels in SubtiExpress: leuB | |
| Metabolic function and regulation of this protein in SubtiPathways: leuB | |
| MW, pI | 39 kDa, 4.744 |
| Gene length, protein length | 1095 bp, 365 aa |
| Immediate neighbours | leuC, leuA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 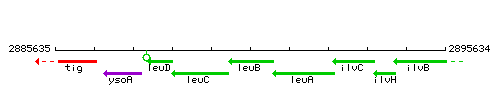
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids, phosphoproteins, most abundant proteins
This gene is a member of the following regulons
CcpA regulon, CodY regulon, T-box, TnrA regulon
The gene
Basic information
- Locus tag: BSU28270
Phenotypes of a mutant
Database entries
- BsubCyc: BSU28270
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: (2R,3S)-3-isopropylmalate + NAD+ = 4-methyl-2-oxopentanoate + CO2 + NADH (according to Swiss-Prot)
- Protein family: LeuB type 1 subfamily (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Modification:
- phosphorylated on Arg-4 PubMed
- Effectors of protein activity:
Database entries
- BsubCyc: BSU28270
- Structure: 1XAD (Thermus thermophilus)
- UniProt: P05645
- KEGG entry: [3]
- E.C. number: 1.1.1.85
Additional information
Expression and regulation
- Regulation:
- for a complete overview on the regulation of the ilv operon, see Brinsmade et al.
- repressed by casamino acids PubMed
- expression is stimulated in the presence of glucose PubMed
- repressed in the absence of good nitrogen sources (glutamine or ammonium) (TnrA) PubMed
- repressed during growth in the presence of branched chain amino acids (CodY) PubMed
- Regulatory mechanism:
- Additional information:
- belongs to the 100 most abundant proteins PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium): 6438 PubMed
- number of protein molecules per cell (complex medium with amino acids, without glucose): 942 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 5218 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 2316 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 1868 PubMed
Biological materials
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References