GlyA
- Description: serine hydroxymethyltransferase
| Gene name | glyA |
| Synonyms | glyC, ipc-34d |
| Essential | yes PubMed |
| Product | serine hydroxymethyltransferase |
| Function | biosynthesis of glycine |
| Gene expression levels in SubtiExpress: glyA | |
| Metabolic function and regulation of this protein in SubtiPathways: glyA | |
| MW, pI | 45 kDa, 5.475 |
| Gene length, protein length | 1245 bp, 415 aa |
| Immediate neighbours | upp, ywlG |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 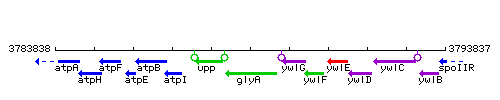
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed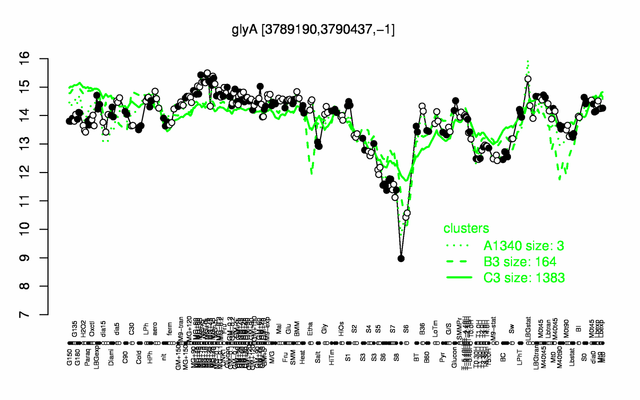
| |
Contents
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids, essential genes, phosphoproteins, most abundant proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU36900
Phenotypes of a mutant
essential PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 5,10-methylenetetrahydrofolate + glycine + H2O = tetrahydrofolate + L-serine (according to Swiss-Prot)
- Protein family: SHMT family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Effectors of protein activity:
- Localization: cytoplasm (according to Swiss-Prot)
Database entries
- Structure: 2VGU (complex with L-serine, Geobacillus stearothermophilus), 2VI8 (Geobacillus stearothermophilus)
- UniProt: P39148
- KEGG entry: [3]
- E.C. number: 2.1.2.1
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- T-box: RNA switch, transcriptional antitermination PubMed
- PurR: transcription repression (molecular inducer: PRPP) PubMed
- Additional information:
- belongs to the 100 most abundant proteins PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Anant Narayan Bhatt, Vinod Bhakuni, Ashutosh Kumar, M Yahiya Khan, Mohammad Imran Siddiqi
Alkaline pH-dependent differential unfolding characteristics of mesophilic and thermophilic homologs of dimeric serine hydroxymethyltransferase.
Biochim Biophys Acta: 2010, 1804(6);1294-300
[PubMed:20152942]
[WorldCat.org]
[DOI]
(P p)
Ana Gutiérrez-Preciado, Tina M Henkin, Frank J Grundy, Charles Yanofsky, Enrique Merino
Biochemical features and functional implications of the RNA-based T-box regulatory mechanism.
Microbiol Mol Biol Rev: 2009, 73(1);36-61
[PubMed:19258532]
[WorldCat.org]
[DOI]
(I p)
Yann Duroc, Carmela Giglione, Thierry Meinnel
Mutations in three distinct loci cause resistance to peptide deformylase inhibitors in Bacillus subtilis.
Antimicrob Agents Chemother: 2009, 53(4);1673-8
[PubMed:19171795]
[WorldCat.org]
[DOI]
(I p)
Christine Eymann, Dörte Becher, Jörg Bernhardt, Katrin Gronau, Anja Klutzny, Michael Hecker
Dynamics of protein phosphorylation on Ser/Thr/Tyr in Bacillus subtilis.
Proteomics: 2007, 7(19);3509-26
[PubMed:17726680]
[WorldCat.org]
[DOI]
(P p)
Alain Lévine, Françoise Vannier, Cédric Absalon, Lauriane Kuhn, Peter Jackson, Elaine Scrivener, Valérie Labas, Joëlle Vinh, Patrick Courtney, Jérôme Garin, Simone J Séror
Analysis of the dynamic Bacillus subtilis Ser/Thr/Tyr phosphoproteome implicated in a wide variety of cellular processes.
Proteomics: 2006, 6(7);2157-73
[PubMed:16493705]
[WorldCat.org]
[DOI]
(P p)
Christine Eymann, Annette Dreisbach, Dirk Albrecht, Jörg Bernhardt, Dörte Becher, Sandy Gentner, Le Thi Tam, Knut Büttner, Gerrit Buurman, Christian Scharf, Simone Venz, Uwe Völker, Michael Hecker
A comprehensive proteome map of growing Bacillus subtilis cells.
Proteomics: 2004, 4(10);2849-76
[PubMed:15378759]
[WorldCat.org]
[DOI]
(P p)
H H Saxild, K Brunstedt, K I Nielsen, H Jarmer, P Nygaard
Definition of the Bacillus subtilis PurR operator using genetic and bioinformatic tools and expansion of the PurR regulon with glyA, guaC, pbuG, xpt-pbuX, yqhZ-folD, and pbuO.
J Bacteriol: 2001, 183(21);6175-83
[PubMed:11591660]
[WorldCat.org]
[DOI]
(P p)