ThrZ
Revision as of 11:54, 7 January 2014 by 134.76.70.252 (talk)
- Description: threonyl-tRNA synthetase (minor)
| Gene name | thrZ |
| Synonyms | thrS2 |
| Essential | no |
| Product | threonyl-tRNA synthetase (minor) |
| Function | translation |
| Gene expression levels in SubtiExpress: thrZ | |
| Metabolic function and regulation of this protein in SubtiPathways: thrZ | |
| MW, pI | 73 kDa, 5.753 |
| Gene length, protein length | 1914 bp, 638 aa |
| Immediate neighbours | ywhA, mmr |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 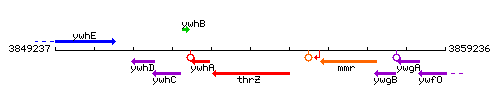
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed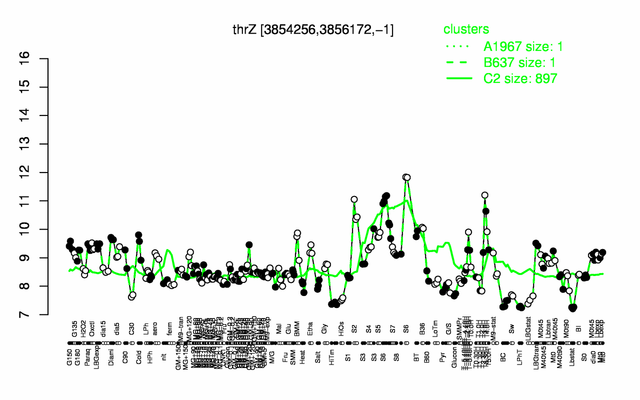
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU37560
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATP + L-threonine + tRNA(Thr) = AMP + diphosphate + L-threonyl-tRNA(Thr) (according to Swiss-Prot)
- Protein family: class-II aminoacyl-tRNA synthetase family (according to Swiss-Prot)
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- cytoplasm (according to Swiss-Prot)
Database entries
- UniProt: P18256
- KEGG entry: [3]
- E.C. number: 6.1.1.3
Additional information
- subject to Clp-dependent proteolysis upon glucose starvation PubMed
Expression and regulation
- Regulatory mechanism:
- T-box: RNA switch, transcriptional antitermination PubMed
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References