YkuP
Revision as of 09:24, 17 April 2014 by 134.76.70.252 (talk)
- Description: flavodoxin, binds FMN, replaces ferredoxin under conditions of iron limitation
| Gene name | ykuP |
| Synonyms | |
| Essential | no |
| Product | flavodoxin |
| Function | electron transfer |
| Gene expression levels in SubtiExpress: ykuP | |
| Interactions involving this protein in SubtInteract: YkuP | |
| Metabolic function and regulation of this protein in SubtiPathways: ykuP | |
| MW, pI | 20 kDa, 4.098 |
| Gene length, protein length | 534 bp, 178 aa |
| Immediate neighbours | ykuO, ykuQ |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed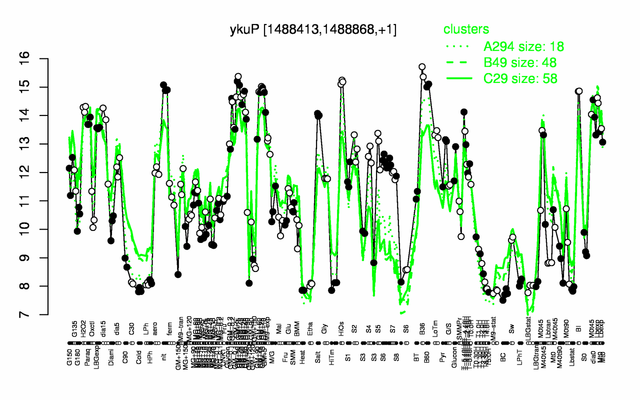
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU14170
Phenotypes of a mutant
Database entries
- BsubCyc: BSU14170
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: flavodoxin-like domain (according to Swiss-Prot)
- Paralogous protein(s): YkuN
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- BsubCyc: BSU14170
- Structure:
- UniProt: O34589
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Additional publications: PubMed