Difference between revisions of "TrpB"
(→Original publications) |
|||
| Line 149: | Line 149: | ||
<pubmed> 18486479 11893063 11163353 7900177 19387555 </pubmed> | <pubmed> 18486479 11893063 11163353 7900177 19387555 </pubmed> | ||
==Original publications== | ==Original publications== | ||
| − | + | <pubmed> 14976255, 3924737, 6436812,2422155,8419914, 1551827, 17507374 21815947 </pubmed> | |
| − | |||
| − | |||
| − | |||
| − | <pubmed> 14976255, 3924737, 6436812,2422155,8419914, 1551827, 17507374 </pubmed> | ||
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 16:23, 13 July 2013
- Description: tryptophan synthase (beta subunit)
| Gene name | trpB |
| Synonyms | |
| Essential | no |
| Product | tryptophan synthase (beta subunit) |
| Function | biosynthesis of tryptophan |
| Gene expression levels in SubtiExpress: trpB | |
| Interactions involving this protein in SubtInteract: TrpB | |
| Metabolic function and regulation of this protein in SubtiPathways: Phe, Tyr, Trp | |
| MW, pI | 43 kDa, 5.526 |
| Gene length, protein length | 1200 bp, 400 aa |
| Immediate neighbours | trpA, trpF |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 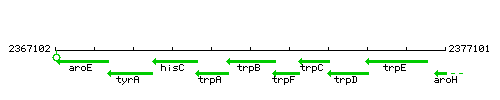
This image was kindly provided by SubtiList
| |
| Expression at a glance PubMed 500px | |
Contents
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU22640
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: L-serine + 1-C-(indol-3-yl)glycerol 3-phosphate = L-tryptophan + glyceraldehyde 3-phosphate + H2O (according to Swiss-Prot)
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure:
- UniProt: P07600
- KEGG entry: [3]
- E.C. number: 4.2.1.20
Additional information
Expression and regulation
- Operon:
- Regulatory mechanism:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications