Difference between revisions of "Sda"
| Line 32: | Line 32: | ||
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
|- | |- | ||
| − | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=sda_2647456_2647614_1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:sda_expression.png|500px]] | + | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=sda_2647456_2647614_1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:sda_expression.png|500px|link=http://subtiwiki.uni-goettingen.de/apps/expression/expression.php?search=BSU25690]] |
|- | |- | ||
|} | |} | ||
Revision as of 13:59, 16 May 2013
- Description: developmental checkpoint protein, inhibits the autokinase activity of KinA and KinB, control of the phosphorelay
| Gene name | sda |
| Synonyms | |
| Essential | no |
| Product | developmental checkpoint protein |
| Function | mediates a developmental checkpoint coupling initiation of sporulation (phosphorylation of Spo0A) to the function of replication initiation proteins |
| Gene expression levels in SubtiExpress: sda | |
| Interactions involving this protein in SubtInteract: Sda | |
| Function and regulation of this protein in SubtiPathways: Phosphorelay | |
| MW, pI | 6 kDa, 5.605 |
| Gene length, protein length | 156 bp, 52 aa |
| Immediate neighbours | yloV, yqeF |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed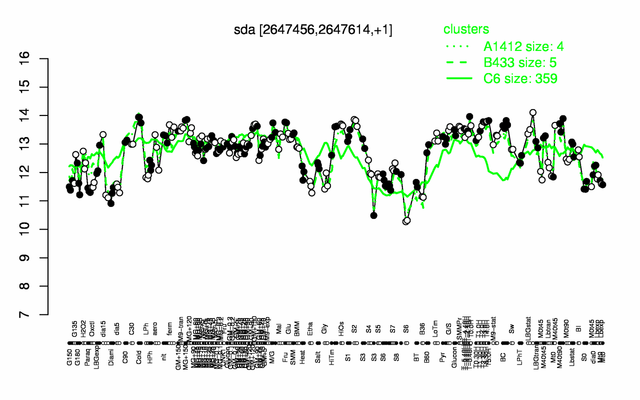
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU25690
Phenotypes of a mutant
- sporulates at a higher frequency
- will sporulate in the presence of replication stress
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Sda is a checkpoint protein that is upregulated in response to replication stress PubMed. Sda inhibits the autokinase activity of KinA (and likely KinB as well). This in its turn results in reduced levels of Spo0A~P. Thus, replication stressed cells will not initiate sporulation.
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- UniProt: Q7WY62
- KEGG entry: [3]
- E.C. number:
Additional information
Sda is rapidly turned over by the ClpP/X system because of some uncharged residues at its C-terminus PubMed.
Expression and regulation
- Regulatory mechanism:
- Additional information:
- Sda is rapidly turned over by the ClpP/X system because of some uncharged residues at its C-terminus PubMed.
- An antisense RNA is predicted for sda PubMed
Biological materials
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References