Difference between revisions of "MutTA"
| Line 35: | Line 35: | ||
<br/><br/><br/><br/> | <br/><br/><br/><br/> | ||
<br/><br/><br/><br/> | <br/><br/><br/><br/> | ||
| − | |||
| − | |||
| − | |||
| − | |||
<br/><br/> | <br/><br/> | ||
| Line 74: | Line 70: | ||
* '''Catalyzed reaction/ biological activity:''' | * '''Catalyzed reaction/ biological activity:''' | ||
** removal of pyrophosphate from the 5' end of mRNAs to make the RNA accessible for degradation by [[RNases]] {{PubMed|21925382}} | ** removal of pyrophosphate from the 5' end of mRNAs to make the RNA accessible for degradation by [[RNases]] {{PubMed|21925382}} | ||
| + | ** RppH requires at least two unpaired nucleotides at the 5' end of its RNA substrates and prefers three or more. The second of these 5'-terminal nucleotides must be G, whereas a less strict preference for a purine is evident at the third position, and A is slightly favored over G at the first position {{PubMed|23610425,23610407}} | ||
* '''Protein family:''' [[Nudix hydrolase]] family (according to Swiss-Prot) | * '''Protein family:''' [[Nudix hydrolase]] family (according to Swiss-Prot) | ||
| Line 97: | Line 94: | ||
=== Database entries === | === Database entries === | ||
| − | * '''Structure:''' | + | * '''Structure:''' [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=4JZV 4JZV] {{PubMed|23610407}} |
* '''UniProt:''' [http://www.uniprot.org/uniprot/O35013 O35013] | * '''UniProt:''' [http://www.uniprot.org/uniprot/O35013 O35013] | ||
| Line 113: | Line 110: | ||
* '''Expression browser:''' [http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=ytkD_3134983_3135459_-1 mutTA] {{PubMed|22383849}} | * '''Expression browser:''' [http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=ytkD_3134983_3135459_-1 mutTA] {{PubMed|22383849}} | ||
| − | * '''Sigma factor:''' [[SigA]] {{PubMed|14761999}}, [[SigF]] {{PubMed|14761999}} | + | * '''[[Sigma factor]]:''' [[SigA]] {{PubMed|14761999}}, [[SigF]] {{PubMed|14761999}} |
* '''Regulation:''' | * '''Regulation:''' | ||
| Line 142: | Line 139: | ||
=References= | =References= | ||
== Reviews == | == Reviews == | ||
| − | + | <pubmed> 22568516</pubmed> | |
| − | |||
| − | |||
| − | |||
| − | |||
== Original publications == | == Original publications == | ||
| − | <pubmed>16513759,16497325, 14761999, 19011023 9387221 15576788 21925382 </pubmed> | + | <pubmed>16513759,16497325, 14761999, 19011023 9387221 15576788 21925382 23610425 23610407 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 07:42, 25 April 2013
- Description: error prevention oxidized guanine system, confers protection against oxidative stress to vegetative cells, moreover the protein has RNA pyrophosphohydrolase activity
| Gene name | mutTA |
| Synonyms | ytkD, rppH |
| Essential | no |
| Product | 8-oxo-dGTPase (antimutator), RNA pyrophosphohydrolase |
| Function | DNA repair, RNA degradation |
| Gene expression levels in SubtiExpress: mutTA | |
| MW, pI | 18 kDa, 5.952 |
| Gene length, protein length | 474 bp, 158 aa |
| Immediate neighbours | ytlD, ytkC |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 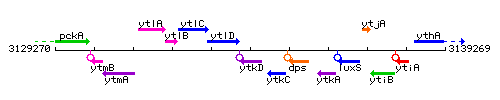
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
DNA repair/ recombination, sporulation proteins, RNases
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU30630
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- removal of pyrophosphate from the 5' end of mRNAs to make the RNA accessible for degradation by RNases PubMed
- RppH requires at least two unpaired nucleotides at the 5' end of its RNA substrates and prefers three or more. The second of these 5'-terminal nucleotides must be G, whereas a less strict preference for a purine is evident at the third position, and A is slightly favored over G at the first position PubMed
- Protein family: Nudix hydrolase family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- UniProt: O35013
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications