Difference between revisions of "PpiB"
| Line 24: | Line 24: | ||
|colspan="2" | '''Genetic context''' <br/> [[Image:ppiB_context.gif]] | |colspan="2" | '''Genetic context''' <br/> [[Image:ppiB_context.gif]] | ||
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
| + | |- | ||
| + | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=ppiB_2435360_2435791_-1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:ppiB_expression.png|500px]] | ||
|- | |- | ||
|} | |} | ||
__TOC__ | __TOC__ | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | |||
<br/><br/> | <br/><br/> | ||
Revision as of 12:31, 19 April 2012
- Description: peptidyl-prolyl isomerase
| Gene name | ppiB |
| Synonyms | cypBS |
| Essential | no |
| Product | peptidyl-prolyl isomerase |
| Function | protein folding |
| MW, pI | 15 kDa, 5.472 |
| Gene length, protein length | 429 bp, 143 aa |
| Immediate neighbours | ypzD, ypuA |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 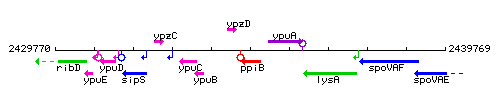
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed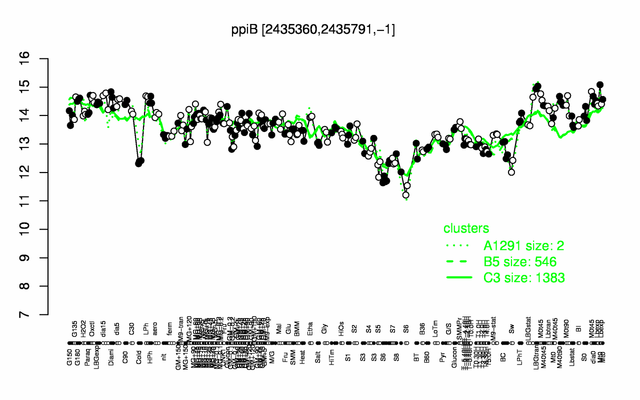
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU23360
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Peptidylproline (omega=180) = peptidylproline (omega=0) (according to Swiss-Prot)
- Protein family: cyclophilin-type PPIase family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure:
- UniProt: P35137
- KEGG entry: [3]
- E.C. number: 5.2.1.8
Additional information
Expression and regulation
- Operon: ppiB PubMed
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References