Difference between revisions of "PtkA"
(→Basic information/ Evolution) |
|||
| Line 103: | Line 103: | ||
* '''Operon:''' ''[[tkmA]]-[[ptkA]]-[[ptpZ]]-[[ugd]]'' {{PubMed|20815827,12970183}} | * '''Operon:''' ''[[tkmA]]-[[ptkA]]-[[ptpZ]]-[[ugd]]'' {{PubMed|20815827,12970183}} | ||
| − | * '''[ | + | * '''Expression browser:''' [http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=ptkA_3731822_3732535_-1 ptkA] {{PubMed|22383849}} |
| + | |||
| + | * '''Sigma factor:''' [[SigA]] {{PubMed|20815827}} | ||
* '''Regulation:''' | * '''Regulation:''' | ||
Revision as of 08:01, 17 April 2012
- Description: protein tyrosine kinase
| Gene name | ptkA |
| Synonyms | ywqD |
| Essential | no |
| Product | protein tyrosine kinase |
| Function | protein phosphorylation |
| Interactions involving this protein in SubtInteract: PtkA | |
| MW, pI | 25 kDa, 9.628 |
| Gene length, protein length | 711 bp, 237 aa |
| Immediate neighbours | ptpZ, tkmA |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 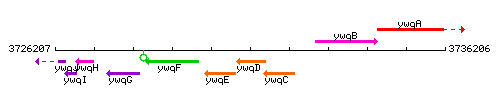
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
biofilm formation, protein modification, membrane proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU36250
Phenotypes of a mutant
- Accumulation of extra chromosome equivalents PubMed
- Defect in biofilm formation, this involves the kinase activity, but the target protein is unknown PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATP + a [protein]-L-tyrosine = ADP + a [protein]-L-tyrosine phosphate (according to Swiss-Prot), autophosphorylation, phosphorylation of Ugd, TuaD, Ssb, SsbB
- Protein family: BY-kinase, see the Bacterial Protein Tyrosine Kinase Database)
- Paralogous protein(s): EpsB
Extended information on the protein
- Kinetic information:
- Domains: single BY-kinase domain
- Modification: autophosphorylation at residues Y225, Y227 and Y228 (primary site) PubMed, dephosphorylated by PtpZ PubMed
- Cofactor(s): ATP
- Effectors of protein activity: TkmA - transmembrane modulator, activates PtkA autophosphorylation and substrate phosphorylation PubMed
Database entries
- Structure: 2VED (CapB, the homolog in Staphylococcus aureus)
- UniProt: P96716
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Additional information:
Biological materials
- Mutant: KO strain created with pMUTIN-2, available from Ivan Mijakovic
- Expression vector: pQE-30, N-terminally 6xHis-tagged, available from Ivan Mijakovic
- lacZ fusion: in a KO strain created with pMUTIN-2, available from Ivan Mijakovic
- GFP fusion: CFP-fusion, available from Ivan Mijakovic
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Ivan Mijakovic, Thiverval-Grignon, France
Your additional remarks
References
Reviews
Original publications
Additional publications: PubMed