Difference between revisions of "RnpA"
(→Extended information on the protein) |
|||
| Line 80: | Line 80: | ||
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| − | * '''[[Interactions]]:''' | + | * '''[[SubtInteract|Interactions]]:''' |
** forms dimers {{PubMed|21764917}} | ** forms dimers {{PubMed|21764917}} | ||
** [[RnpA]]-[[RpmH]] | ** [[RnpA]]-[[RpmH]] | ||
| Line 140: | Line 140: | ||
==Original Publications== | ==Original Publications== | ||
'''Additional publications:''' {{PubMed|21076397,20843005,21622899}} | '''Additional publications:''' {{PubMed|21076397,20843005,21622899}} | ||
| − | <pubmed>10390342,17919279,17355991,11812129,16980484,11454206, 19549719,11454206 9563955, 19932118 17299131 20476778 11258888 16185070 </pubmed> | + | <pubmed>10390342,17919279,17355991,11812129,16980484,11454206, 19549719,11454206 9563955, 19932118 17299131 20476778 11258888 16185070 21764917</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 17:36, 19 July 2011
| Gene name | rnpA |
| Synonyms | |
| Essential | yes PubMed |
| Product | protein component of RNase P (substrate specificity) |
| Function | cleavage of precursor sequences from the 5' ends of pre-tRNAs |
| MW, pI | 13 kDa, 10.804 |
| Gene length, protein length | 348 bp, 116 aa |
| Immediate neighbours | spoIIIJ, rpmH |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 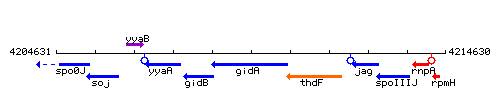
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
Rnases, translation, essential genes
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU41050
Phenotypes of a mutant
essential PubMed
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Endonucleolytic cleavage of RNA, removing 5'-extranucleotides from tRNA precursor (according to Swiss-Prot)
- Protein family: rnpA family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
Database entries
- Structure:
- UniProt: P25814
- KEGG entry: [2]
- E.C. number: 3.1.26.5
Additional information
Expression and regulation
- Operon:
- Sigma factor:
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
- Carol Fierke, University of Michigan, Ann Arbor, USA homepage
- Roland Hartmann, Marburg University, Germany homepage
Your additional remarks
References
Reviews
Original Publications
Additional publications: PubMed