Difference between revisions of "FabF"
| Line 103: | Line 103: | ||
* '''Operon:''' ''[[fabHA]]-[[fabF]]'' {{PubMed|12737802}} | * '''Operon:''' ''[[fabHA]]-[[fabF]]'' {{PubMed|12737802}} | ||
| − | * '''[[Sigma factor]]:''' [[SigA]] {{PubMed|12737802}} | + | * '''[[Sigma factor]]:''' |
| + | ** ''[[fabHA]]'': [[SigA]] {{PubMed|12737802}} | ||
| + | ** ''[[fabF]]'': [[SigW]] {{PubMed|21542858}} | ||
* '''Regulation:''' | * '''Regulation:''' | ||
| − | ** expressed when the cells experience a lack of malonyl-CoA ([[FapR]]) {{PubMed|12737802}} | + | ** ''[[fabHA]]'': expressed when the cells experience a lack of malonyl-CoA ([[FapR]]) {{PubMed|12737802}} |
| − | ** induced upon fatty acid biosynthesis inhibition {{PubMed|21383089}} | + | ** ''[[fabHA]]'': induced upon fatty acid biosynthesis inhibition {{PubMed|21383089}} |
| + | ** ''[[fabF]]'': induced by alkaline shock and by polymyxin B, vancomycin, cephalosporin C, D-cycloserine, and triton X-100 ([[SigW]]) {{PubMed|21542858}} | ||
* '''Regulatory mechanism:''' | * '''Regulatory mechanism:''' | ||
| Line 136: | Line 139: | ||
<pubmed> 15952903 17919287</pubmed> | <pubmed> 15952903 17919287</pubmed> | ||
==Original Publications== | ==Original Publications== | ||
| + | '''Additional publications:''' {{PubMed|21542858}} | ||
<pubmed>18384517, 12837788,12737802, 12682299 21383089</pubmed> | <pubmed>18384517, 12837788,12737802, 12682299 21383089</pubmed> | ||
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 14:29, 6 May 2011
- Description: beta-ketoacyl-acyl carrier protein synthase II
| Gene name | fabF |
| Synonyms | yjaY |
| Essential | yes PubMed |
| Product | beta-ketoacyl-acyl carrier protein synthase II |
| Function | fatty acid biosynthesis |
| Metabolic function and regulation of this protein in SubtiPathways: Lipid synthesis | |
| MW, pI | 43 kDa, 4.768 |
| Gene length, protein length | 1239 bp, 413 aa |
| Immediate neighbours | fabHA, yjaZ |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 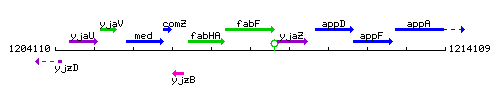
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
biosynthesis of lipids, essential genes
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU11340
Phenotypes of a mutant
essential PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: (Z)-hexadec-11-enoyl-[acyl-carrier-protein] + malonyl-[acyl-carrier-protein] = (Z)-3-oxooctadec-13-enoyl-[acyl-carrier-protein] + CO2 + [acyl-carrier-protein] (according to Swiss-Prot)
- Protein family: beta-ketoacyl-ACP synthases family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization:
Database entries
- UniProt: O34340
- KEGG entry: [3]
- E.C. number: 2.3.1.179
Additional information
Expression and regulation
- Regulation:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original Publications
Additional publications: PubMed
Michaela Wenzel, Malay Patra, Dirk Albrecht, David Y-K Chen, K C Nicolaou, Nils Metzler-Nolte, Julia E Bandow
Proteomic signature of fatty acid biosynthesis inhibition available for in vivo mechanism-of-action studies.
Antimicrob Agents Chemother: 2011, 55(6);2590-6
[PubMed:21383089]
[WorldCat.org]
[DOI]
(I p)
Gustavo E Schujman, Silvia Altabe, Diego de Mendoza
A malonyl-CoA-dependent switch in the bacterial response to a dysfunction of lipid metabolism.
Mol Microbiol: 2008, 68(4);987-96
[PubMed:18384517]
[WorldCat.org]
[DOI]
(I p)
Allen C Price, Charles O Rock, Stephen W White
The 1.3-Angstrom-resolution crystal structure of beta-ketoacyl-acyl carrier protein synthase II from Streptococcus pneumoniae.
J Bacteriol: 2003, 185(14);4136-43
[PubMed:12837788]
[WorldCat.org]
[DOI]
(P p)
Gustavo E Schujman, Luciana Paoletti, Alan D Grossman, Diego de Mendoza
FapR, a bacterial transcription factor involved in global regulation of membrane lipid biosynthesis.
Dev Cell: 2003, 4(5);663-72
[PubMed:12737802]
[WorldCat.org]
[DOI]
(P p)
K Kobayashi, S D Ehrlich, A Albertini, G Amati, K K Andersen, M Arnaud, K Asai, S Ashikaga, S Aymerich, P Bessieres, F Boland, S C Brignell, S Bron, K Bunai, J Chapuis, L C Christiansen, A Danchin, M Débarbouille, E Dervyn, E Deuerling, K Devine, S K Devine, O Dreesen, J Errington, S Fillinger, S J Foster, Y Fujita, A Galizzi, R Gardan, C Eschevins, T Fukushima, K Haga, C R Harwood, M Hecker, D Hosoya, M F Hullo, H Kakeshita, D Karamata, Y Kasahara, F Kawamura, K Koga, P Koski, R Kuwana, D Imamura, M Ishimaru, S Ishikawa, I Ishio, D Le Coq, A Masson, C Mauël, R Meima, R P Mellado, A Moir, S Moriya, E Nagakawa, H Nanamiya, S Nakai, P Nygaard, M Ogura, T Ohanan, M O'Reilly, M O'Rourke, Z Pragai, H M Pooley, G Rapoport, J P Rawlins, L A Rivas, C Rivolta, A Sadaie, Y Sadaie, M Sarvas, T Sato, H H Saxild, E Scanlan, W Schumann, J F M L Seegers, J Sekiguchi, A Sekowska, S J Séror, M Simon, P Stragier, R Studer, H Takamatsu, T Tanaka, M Takeuchi, H B Thomaides, V Vagner, J M van Dijl, K Watabe, A Wipat, H Yamamoto, M Yamamoto, Y Yamamoto, K Yamane, K Yata, K Yoshida, H Yoshikawa, U Zuber, N Ogasawara
Essential Bacillus subtilis genes.
Proc Natl Acad Sci U S A: 2003, 100(8);4678-83
[PubMed:12682299]
[WorldCat.org]
[DOI]
(P p)