Difference between revisions of "KinC"
(→References) |
|||
| Line 1: | Line 1: | ||
| − | * '''Description:''' [[two-component systems|two-component]] sensor kinase, phosphorylates [[Spo0F]] and [[Spo0A]] | + | * '''Description:''' [[two-component systems|two-component]] sensor kinase, phosphorylates [[Spo0F]] and [[Spo0A]], part of the [[phosphorelay]], governs expression of genes involved in [[biofilm formation]] <br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
| Line 39: | Line 39: | ||
<br/><br/><br/><br/> | <br/><br/><br/><br/> | ||
<br/><br/><br/><br/> | <br/><br/><br/><br/> | ||
| − | + | <br/><br/> | |
| − | |||
= [[Categories]] containing this gene/protein = | = [[Categories]] containing this gene/protein = | ||
| Line 79: | Line 78: | ||
** autophosphorylation, phosphorylation of [[Spo0F]] as part of the [[phosphorelay]], but also direct phosphorylation of [[Spo0A]] {{PubMed|19114652}} | ** autophosphorylation, phosphorylation of [[Spo0F]] as part of the [[phosphorelay]], but also direct phosphorylation of [[Spo0A]] {{PubMed|19114652}} | ||
** mainly active in the younger, outer regions of a colony (with [[KinD]]) {{PubMed|21097618}} | ** mainly active in the younger, outer regions of a colony (with [[KinD]]) {{PubMed|21097618}} | ||
| − | ** phosphorylates [[Spo0A]] in response to the presence of surfactin {{PubMed|22882210}} | + | ** phosphorylates [[Spo0A]] in response to the presence of surfactin {{PubMed|22882210}}, this has been refuted {{PubMed|25701730}} |
* '''Protein family:''' | * '''Protein family:''' | ||
| Line 99: | Line 98: | ||
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| − | ** activity is triggered by potassium leakage {{PubMed|19114652}} | + | ** activity is triggered by potassium leakage {{PubMed|19114652}}, this has been refuted {{PubMed|25701730}} |
** activity is triggered by polyisoprenoid lipids formed by [[YisP]] {{PubMed|20713508}} | ** activity is triggered by polyisoprenoid lipids formed by [[YisP]] {{PubMed|20713508}} | ||
| Line 109: | Line 108: | ||
** cell membrane (Heterogeneous) [http://www.ncbi.nlm.nih.gov/sites/entrez/16479537 PubMed] | ** cell membrane (Heterogeneous) [http://www.ncbi.nlm.nih.gov/sites/entrez/16479537 PubMed] | ||
** co-localizes with [[FloT]] in discrete foci in the membrane {{PubMed|20713508}} | ** co-localizes with [[FloT]] in discrete foci in the membrane {{PubMed|20713508}} | ||
| − | ** the localization of [[KinC]] in membrane microdomains depends on [[FloA]] and [[FloT]] {{PubMed|22882210}} | + | ** the localization of [[KinC]] in membrane microdomains depends on [[FloA]] and [[FloT]] {{PubMed|22882210}}, this has been refuted {{PubMed|25701730}} |
=== Database entries === | === Database entries === | ||
| Line 163: | Line 162: | ||
<pubmed> 25652542</pubmed> | <pubmed> 25652542</pubmed> | ||
== Original publications == | == Original publications == | ||
| − | <pubmed>19114652,10094672,11069677,16166384, 20713508,8002615, 16479537 8002614 20946851 20971918 21097618 22882210 23927765 </pubmed> | + | <pubmed>19114652,10094672,11069677,16166384, 20713508,8002615, 16479537 8002614 20946851 20971918 21097618 22882210 23927765 25701730</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 17:02, 23 February 2015
- Description: two-component sensor kinase, phosphorylates Spo0F and Spo0A, part of the phosphorelay, governs expression of genes involved in biofilm formation
| Gene name | kinC |
| Synonyms | ssb |
| Essential | no |
| Product | two-component sensor kinase |
| Function | initiation of sporulation |
| Gene expression levels in SubtiExpress: kinC | |
| Interactions involving this protein in SubtInteract: KinC | |
| Function and regulation of this protein in SubtiPathways: kinC | |
| MW, pI | 48 kDa, 6.225 |
| Gene length, protein length | 1284 bp, 428 aa |
| Immediate neighbours | abh, ykqA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 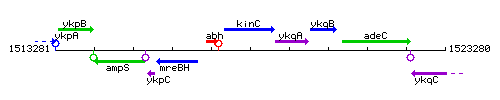
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
protein modification, transcription factors and their control, phosphorelay, biofilm formation, membrane proteins, phosphoproteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU14490
Phenotypes of a mutant
- defective in biofilm formation PubMed
Database entries
- BsubCyc: BSU14490
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- two transmembrane segments
- PAS domain
- C-terminal histidine phosphotransferase domain
- Modification: autophosphorylation on a His residue
- Cofactor(s):
- Effectors of protein activity:
Database entries
- BsubCyc: BSU14490
- Structure:
- UniProt: P39764
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: kinC (according to DBTBS)
- Additional information:
Biological materials
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications