Difference between revisions of "HtrA"
| Line 79: | Line 79: | ||
* '''Kinetic information:''' | * '''Kinetic information:''' | ||
| − | * '''Domains:''' | + | * '''[[Domains]]:''' |
* '''Modification:''' | * '''Modification:''' | ||
| − | * ''' | + | * '''[[Cofactors]]:''' |
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| Line 112: | Line 112: | ||
* '''Expression browser:''' [http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=htrA_1357936_1359285_-1 htrA] {{PubMed|22383849}} | * '''Expression browser:''' [http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=htrA_1357936_1359285_-1 htrA] {{PubMed|22383849}} | ||
| − | * '''Sigma factor:''' [[SigA]] {{PubMed|10692364}} | + | * '''[[Sigma factor]]:''' [[SigA]] {{PubMed|10692364}} |
* '''Regulation:''' | * '''Regulation:''' | ||
| Line 122: | Line 122: | ||
* '''Additional information:''' | * '''Additional information:''' | ||
| + | ** [[HtrA]] is subject to degradation by [[WprA]] and other extracellular proteases {{PubMed|24362423}} | ||
=Biological materials = | =Biological materials = | ||
| Line 147: | Line 148: | ||
== Original publications == | == Original publications == | ||
| − | <pubmed>17600057,12270824 ,11555295,18957862, 10692364 21624103 22307758 22092710</pubmed> | + | <pubmed>17600057,12270824 ,11555295,18957862, 10692364 21624103 22307758 22092710 24362423</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 16:29, 16 January 2014
- Description: membrane-anchored protein quality control protease, serine protease Do
| Gene name | htrA |
| Synonyms | ykdA |
| Essential | no |
| Product | serine protease Do |
| Function | protein quality control |
| Gene expression levels in SubtiExpress: htrA | |
| Metabolic function and regulation of this protein in SubtiPathways: htrA | |
| MW, pI | 47 kDa, 4.699 |
| Gene length, protein length | 1347 bp, 449 aa |
| Immediate neighbours | ykcC, proG |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 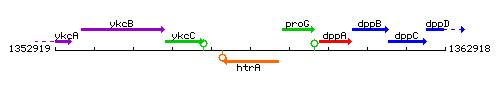
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed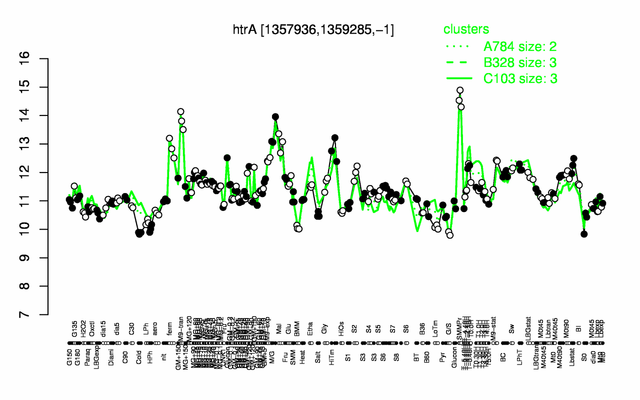
| |
Contents
Categories containing this gene/protein
proteolysis, heat shock proteins, membrane proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU12900
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: PDZ (DHR) domain (according to Swiss-Prot)
- Paralogous protein(s): HtrC
Extended information on the protein
- Kinetic information:
- Modification:
- Effectors of protein activity:
Database entries
- Structure:
- UniProt: O34358
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: htrA (according to DBTBS)
- Regulation:
- Additional information:
Biological materials
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications