Difference between revisions of "LytD"
| Line 76: | Line 76: | ||
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| − | * '''Interactions:''' | + | * '''[[SubtInteract|Interactions]]:''' |
| − | * '''Localization:''' | + | * '''[[Localization]]:''' |
| + | ** extracellular (signal peptide) [http://www.ncbi.nlm.nih.gov/pubmed/18957862 PubMed] | ||
=== Database entries === | === Database entries === | ||
| Line 124: | Line 125: | ||
=References= | =References= | ||
| − | <pubmed>7934877,19542270 ,18957862, </pubmed> | + | <pubmed>7934877,19542270 ,18957862, 21821766</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 11:34, 15 August 2011
- Description: glucosaminidase
| Gene name | lytD |
| Synonyms | cwlG |
| Essential | no |
| Product | glucosaminidase |
| Function | major autolysin, cell separation |
| MW, pI | 95 kDa, 9.935 |
| Gene length, protein length | 2640 bp, 880 aa |
| Immediate neighbours | tagC, pmi |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 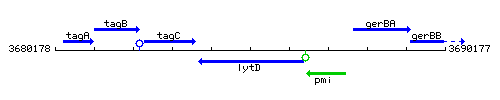
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
cell wall degradation/ turnover
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU35780
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
- A mutation was found in this gene after evolution under relexed selection for sporulation PubMed
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Endohydrolysis of the N,N'-diacetylchitobiosyl unit in high-mannose glycopeptides and glycoproteins containing the -(Man(GlcNAc)2)Asn-structure (according to Swiss-Prot)
- Protein family: glycosyl hydrolase 73 family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- extracellular (signal peptide) PubMed
Database entries
- Structure:
- UniProt: P39848
- KEGG entry: [3]
- E.C. number: 3.2.1.96
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References