Difference between revisions of "PtsG"
| Line 127: | Line 127: | ||
<pubmed>10627040 ,12850135 ,18763711 ,11902727 ,9765562 ,9513271 ,1956301 ,10543968 ,17074746 ,15155854 ,14527945 ,1508157 ,2120236 </pubmed> | <pubmed>10627040 ,12850135 ,18763711 ,11902727 ,9765562 ,9513271 ,1956301 ,10543968 ,17074746 ,15155854 ,14527945 ,1508157 ,2120236 </pubmed> | ||
| + | |||
| + | [[Category:Protein-coding genes]] | ||
Revision as of 09:53, 21 July 2009
- Description: trigger enzyme: major glucose permease of the PTS, EIICBA(Glc) and control of GlcT activity
| Gene name | ptsG |
| Synonyms | ptsX, crr |
| Essential | no |
| Product | glucose-specific enzyme IICBA component |
| Function | glucose transport and phosphorylation, control of GlcT activity |
| Metabolic function and regulation of this protein in SubtiPathways: Central C-metabolism, Sugar catabolism | |
| MW, pI | 75,3 kDa, 5.40 |
| Gene length, protein length | 2097 bp, 699 amino acids |
| Immediate neighbours | glcT, ptsH |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 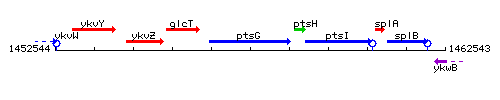
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU13890
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: transport and phosphorylation of glucose, receives a phosphate from HPr at the IIA domain (His-620), the phosphate group is then transferred to the IIB domain (Cys-461) an finally to the incoming glucose. In the absence of glucose, PtsG phosphorylates and thereby inactivates the transcriptional antiterminator GlcT.
- Protein family: PTS permease, glucose permease (Glc) family PubMed, PTS enzyme II, glucose family
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- 11x transmembrane domain (16–36, 89–109, 139–159, 180–200, 233–253, 283–303, 313–333, 338–358, 365–385, 388–408)
- PTS EIIC domain ( 1-424)
- PTS EIIB domain (439–520)
- PTS EIIA domain (568–672)
- Modification: transient phosphorylation (HPr-dependent) on His-620, then internal phosphotransfer from His-620 to Cys-461
- Cofactor(s):
- Effectors of protein activity:
- Localization: membrane protein PubMed
Database entries
- UniProt: P20166
- KEGG entry: [3]
- E.C. number: 2.7.1.69 9 2.7.1.69]
Additional information
Expression and regulation
- Regulation: expression activated by glucose (32 fold) PubMed
- Regulatory mechanism: transcriptional antitermination via the GlcT-dependent RNA-switch PubMed
- Additional information:
Biological materials
- Mutant: GP474 (cat), QB5436 (spc), QB5445 (erm), available in Stülke lab
- Expression vector: pGP123 (domains BA, in pWH844), pGP123 (domains BA, mut: H620D, in pWH844), pGP428 (EIIB, in pWH844), pGP437(EIIA in pWH844, with thrombin cleavage site), available in Stülke lab
- lacZ fusion: pGP34 (pAC5), pGP66 (pAC7), pGP606 (mutant terminator, pAC6), pGP532 (pAC7), series of promoter deletions are available in pAC5 and pAC6, series of RAT mutants are available in pAC6, available in Stülke lab
- GFP fusion:
- Antibody:
Labs working on this gene/protein
Jörg Stülke, University of Göttingen, Germany Homepage
Your additional remarks
References