Difference between revisions of "Ung"
| Line 58: | Line 58: | ||
* increased mutation rates {{PubMed|22056936}} | * increased mutation rates {{PubMed|22056936}} | ||
=== Database entries === | === Database entries === | ||
| + | * '''BsubCyc:''' [http://bsubcyc.org/BSUB/NEW-IMAGE?type=NIL&object=BSU37970&redirect=T BSU37970] | ||
* '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ung.html] | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ung.html] | ||
| Line 93: | Line 94: | ||
=== Database entries === | === Database entries === | ||
| + | * '''BsubCyc:''' [http://bsubcyc.org/BSUB/NEW-IMAGE?type=NIL&object=BSU37970&redirect=T BSU37970] | ||
* '''Structure:''' [http://www.rcsb.org/pdb/explore/explore.do?pdbId=3A7N 3A7N] (from ''Mycobacterium tuberculosis'', 42% identity) {{PubMed|20693660}} | * '''Structure:''' [http://www.rcsb.org/pdb/explore/explore.do?pdbId=3A7N 3A7N] (from ''Mycobacterium tuberculosis'', 42% identity) {{PubMed|20693660}} | ||
Revision as of 15:07, 2 April 2014
- Description: uracil-DNA glycosylase
| Gene name | ung |
| Synonyms | ipa-57d, ywdG |
| Essential | no |
| Product | uracil-DNA glycosylase |
| Function | DNA repair |
| Gene expression levels in SubtiExpress: ung | |
| Interactions involving this protein in SubtInteract: Ung | |
| MW, pI | 25 kDa, 8.782 |
| Gene length, protein length | 675 bp, 225 aa |
| Immediate neighbours | ywdH, ywdF |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 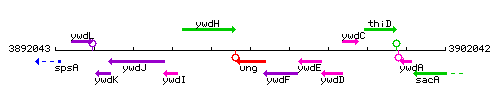
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed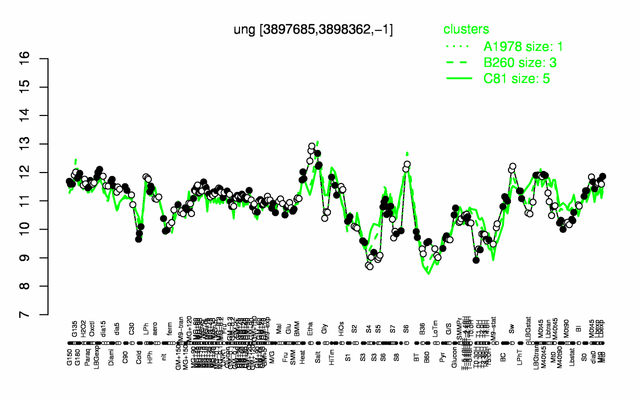
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU37970
Phenotypes of a mutant
- increased mutation rates PubMed
Database entries
- BsubCyc: BSU37970
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: removes uracil preferentially from single-stranded DNA over double-stranded DNA, exhibiting higher preference for U:G than U:A mismatches PubMed
- Protein family: uracil-DNA glycosylase family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: cytoplasm (according to Swiss-Prot)
Database entries
- BsubCyc: BSU37970
- UniProt: P39615
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: ung PubMed
- Sigma factor:
- Regulation:
- expressed throughout growth and staionary phase PubMed
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Original publications
Karina López-Olmos, Martha P Hernández, Jorge A Contreras-Garduño, Eduardo A Robleto, Peter Setlow, Ronald E Yasbin, Mario Pedraza-Reyes
Roles of endonuclease V, uracil-DNA glycosylase, and mismatch repair in Bacillus subtilis DNA base-deamination-induced mutagenesis.
J Bacteriol: 2012, 194(2);243-52
[PubMed:22056936]
[WorldCat.org]
[DOI]
(I p)
Laura Pérez-Lago, Gemma Serrano-Heras, Benito Baños, José M Lázaro, Martín Alcorlo, Laurentino Villar, Margarita Salas
Characterization of Bacillus subtilis uracil-DNA glycosylase and its inhibition by phage φ29 protein p56.
Mol Microbiol: 2011, 80(6);1657-66
[PubMed:21542855]
[WorldCat.org]
[DOI]
(I p)
Audrey Costes, François Lecointe, Stephen McGovern, Sophie Quevillon-Cheruel, Patrice Polard
The C-terminal domain of the bacterial SSB protein acts as a DNA maintenance hub at active chromosome replication forks.
PLoS Genet: 2010, 6(12);e1001238
[PubMed:21170359]
[WorldCat.org]
[DOI]
(I e)
Prem Singh Kaushal, Ramappa K Talawar, Umesh Varshney, M Vijayan
Structure of uracil-DNA glycosylase from Mycobacterium tuberculosis: insights into interactions with ligands.
Acta Crystallogr Sect F Struct Biol Cryst Commun: 2010, 66(Pt 8);887-92
[PubMed:20693660]
[WorldCat.org]
[DOI]
(I p)
E Presecan, I Moszer, L Boursier, H Cruz Ramos, V de la Fuente, M-F Hullo, C Lelong, S Schleich, A Sekowska, B H Song, G Villani, F Kunst, A Danchin, P Glaser
The Bacillus subtilis genome from gerBC (311 degrees) to licR (334 degrees).
Microbiology (Reading): 1997, 143 ( Pt 10);3313-3328
[PubMed:9353933]
[WorldCat.org]
[DOI]
(P p)