Difference between revisions of "PbpA"
| Line 29: | Line 29: | ||
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
|- | |- | ||
| − | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=pbpA_2581771_2583921_-1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:pbpA_expression.png|500px]] | + | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=pbpA_2581771_2583921_-1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:pbpA_expression.png|500px|link=http://subtiwiki.uni-goettingen.de/apps/expression/expression.php?search=BSU25000]] |
|- | |- | ||
|} | |} | ||
Revision as of 13:55, 16 May 2013
- Description: penicillin-binding protein PBP 2A
| Gene name | pbpA |
| Synonyms | yqgF |
| Essential | no |
| Product | penicillin-binding protein PBP 2A |
| Function | formation of a rod-shaped peptidoglycan cell wall, spore outgrowth |
| Gene expression levels in SubtiExpress: pbpA | |
| Interactions involving this protein in SubtInteract: PbpA | |
| MW, pI | 79 kDa, 9.571 |
| Gene length, protein length | 2148 bp, 716 aa |
| Immediate neighbours | pstS, yqgE |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 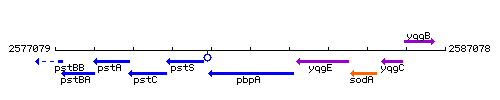
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU25000
Phenotypes of a mutant
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- part of the cell wall biosynthetic complex PubMed
- folding requires PrsA PubMed
- Localization: extracellular (signal peptide) PubMed
Database entries
- Structure:
- UniProt: P54488
- KEGG entry: [2]
- E.C. number:
Additional information
Expression and regulation
- Sigma factor:
- Regulation:
- Regulatory mechanism:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Jeff Errington lab
- Antibody:
Labs working on this gene/protein
Jeff Errington, Newcastle University, UK homepage
Your additional remarks
References
Lehnik-Habrink M, Schaffer M, Mäder U, Diethmaier C, Herzberg C, Stülke J RNA processing in Bacillus subtilis: identification of targets of the essential RNase Y. Mol Microbiol. 2011 81(6): 1459-1473. PubMed:21815947