Difference between revisions of "CshA"
| Line 56: | Line 56: | ||
===Phenotypes of a mutant === | ===Phenotypes of a mutant === | ||
* poor growth at low temperatures (16 to 20°C) {{PubMed|23175651}} | * poor growth at low temperatures (16 to 20°C) {{PubMed|23175651}} | ||
| − | * reduced number of | + | * reduced number of [[ribosome]]s {{PubMed|23175651}} |
=== Database entries === | === Database entries === | ||
| Line 70: | Line 70: | ||
=== Basic information/ Evolution === | === Basic information/ Evolution === | ||
| − | * '''Catalyzed reaction/ biological activity:''' RNA helicase | + | * '''Catalyzed reaction/ biological activity:''' |
| + | ** RNA helicase | ||
| + | ** required for [[ribosome]] assembly (biogenesis of the large subunit) {{PubMed|23175651}} | ||
* '''Protein family:''' helicase C-terminal domain (according to Swiss-Prot) [[DEAD-box RNA helicases|DEAD-box RNA helicase]] | * '''Protein family:''' helicase C-terminal domain (according to Swiss-Prot) [[DEAD-box RNA helicases|DEAD-box RNA helicase]] | ||
| Line 100: | Line 102: | ||
** [[DeaD]]-[[CshA]] {{PubMed|20572937}} | ** [[DeaD]]-[[CshA]] {{PubMed|20572937}} | ||
** [[RnpA]]-[[CshA]] {{PubMed|21764917}} | ** [[RnpA]]-[[CshA]] {{PubMed|21764917}} | ||
| + | ** [[RplA]]-[[CshA]] {{PubMed|23175651}} | ||
| + | ** [[RplC]]-[[CshA]] {{PubMed|23175651}} | ||
* '''[[Localization]]:''' cytoplasma, colocalizes with the ribosomes [http://www.ncbi.nlm.nih.gov/sites/entrez/16352840 PubMed], cell membrane {{PubMed|20572937}} | * '''[[Localization]]:''' cytoplasma, colocalizes with the ribosomes [http://www.ncbi.nlm.nih.gov/sites/entrez/16352840 PubMed], cell membrane {{PubMed|20572937}} | ||
Revision as of 16:48, 26 November 2012
- Description: DEAD-box RNA helicase, important for adaptation to low temperatures
| Gene name | cshA |
| Synonyms | ydbR |
| Essential | no |
| Product | DEAD-box RNA helicase |
| Function | RNA helicase |
| Gene expression levels in SubtiExpress: cshA | |
| Interactions involving this protein in SubtInteract: CshA | |
| MW, pI | 57 kDa, 9.89 |
| Gene length, protein length | 1533 bp, 511 aa |
| Immediate neighbours | murF, ydbS |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 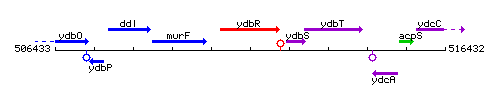
This image was kindly provided by SubtiList
| |
[None Expression at a glance] PubMed
| |
Contents
Categories containing this gene/protein
DEAD-box RNA helicases, translation, cold stress proteins, membrane proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU04580
Phenotypes of a mutant
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: helicase C-terminal domain (according to Swiss-Prot) DEAD-box RNA helicase
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: cytoplasma, colocalizes with the ribosomes PubMed, cell membrane PubMed
Database entries
- Structure:
- UniProt: P96614
- KEGG entry: [2]
- E.C. number:
Additional information
Expression and regulation
- Operon:
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- GP1035 (aphA3), available in Jörg Stülke's lab
- GP1083 (cat), available in Jörg Stülke's lab
- Expression vector:
- for expression/ purification from B. subtilis with C-terminal Strep-tag, for SPINE, in pGP382: pGP1387, available in Jörg Stülke's lab
- for expression/ purification from B. subtilis with C-terminal Strep-tag, for SPINE, expression from the native chromomsomal site: GP1026 (aphA3), available in Jörg Stülke's lab
- for expression/ purification from E. coli with N-terminal His-tag, in pWH844: pGP1386, available in Jörg Stülke's lab
- lacZ fusion:
- GFP fusion:
- pGP1369 for chromosomal expression of CshA-YFP, available in Jörg Stülke's lab
- B. subtilis GP1081 cshA-gfp spc, available in Jörg Stülke's lab,
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Jörg Stülke's lab
- FLAG-tag construct:
- GP1010 (spc, based on pGP1331), available in Jörg Stülke's lab
- GP1074 (tet), available in Jörg Stülke's lab
- Antibody:
Labs working on this gene/protein
Mohamed Marahiel, Marburg University, Germany homepage
Your additional remarks
References
Additional publications: PubMed