Difference between revisions of "DeaD"
| Line 28: | Line 28: | ||
|colspan="2" | '''Genetic context''' <br/> [[Image:deaD_context.gif]] | |colspan="2" | '''Genetic context''' <br/> [[Image:deaD_context.gif]] | ||
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
| + | |- | ||
| + | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=deaD_4015987_4017426_-1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:deaD_expression.png|500px]] | ||
|- | |- | ||
|} | |} | ||
__TOC__ | __TOC__ | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | <br/><br/><br/><br/> | ||
| + | |||
<br/><br/> | <br/><br/> | ||
Revision as of 12:22, 23 April 2012
- Description: DEAD-box RNA helicase
| Gene name | deaD |
| Synonyms | yxiN |
| Essential | no |
| Product | DEAD-box RNA helicase |
| Function | RNA helicase |
| Interactions involving this protein in SubtInteract: DeaD | |
| MW, pI | 53 kDa, 7.706 |
| Gene length, protein length | 1437 bp, 479 aa |
| Immediate neighbours | yxiO, yxiM |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 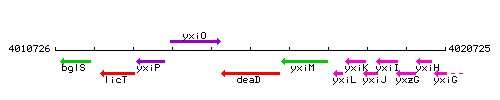
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
DEAD-box RNA helicases, translation
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU39110
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: RNA helicase
- Protein family: DEAD-box RNA helicase
Extended information on the protein
- Kinetic information:
- Domains:
- Helicase domain 1: AS 1-203
- Linker region: AS 204-212
- Helicase domain 2: AS 207-368
- RNA-binding domain: AS 404-479
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure: 2G0C (RNA-binding domain, AA 404-479), 2HJV (second domain, AA 207-368); 3MOJ (RNA binding domain complexed with a fragment of 23S ribosomal RNA) PubMed
- UniProt: P42305
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Sigma factor:
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant: GP1121 (tet), available in the Stülke lab
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Stülke lab
- Antibody:
Labs working on this gene/protein
Dagmar Klostermeier, Biozentrum Basel, Switzerland homepage
Your additional remarks
References