Difference between revisions of "TrpC"
| Line 56: | Line 56: | ||
=== Additional information=== | === Additional information=== | ||
| − | |||
| − | |||
| − | |||
=The protein= | =The protein= | ||
| Line 82: | Line 79: | ||
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| − | * '''Interactions:''' | + | * '''[[SubtInteract|Interactions]]:''' |
| − | * '''Localization:''' | + | * '''[[Localization]]:''' |
=== Database entries === | === Database entries === | ||
| Line 107: | Line 104: | ||
* '''Regulation:''' | * '''Regulation:''' | ||
| − | ** induction by tryptophan ([[MtrB| TRAP]]) {{PubMed|1551827}} | + | ** induction by tryptophan ([[MtrB|TRAP]]) {{PubMed|1551827}} |
* '''Regulatory mechanism:''' | * '''Regulatory mechanism:''' | ||
| − | ** [[MtrB| TRAP]]: binding to the mRNA in the presence of tryptophan, this results in transcription termination {{PubMed|8419914}} | + | ** [[MtrB|TRAP]]: binding to the mRNA in the presence of tryptophan, this results in transcription termination {{PubMed|8419914}} |
* '''Additional information:''' | * '''Additional information:''' | ||
| + | ** the mRNA is substantially stabilized upon depletion of [[Rny|RNase Y]] {{PubMed|21815947}} | ||
=Biological materials = | =Biological materials = | ||
| Line 133: | Line 131: | ||
=References= | =References= | ||
| − | + | <big>''Lehnik-Habrink M, Schaffer M, Mäder U, Diethmaier C, Herzberg C, Stülke J'' </big> | |
| + | <big>'''RNA processing in ''Bacillus subtilis'': identification of targets of the essential RNase Y.''' </big> | ||
| + | <big>Mol Microbiol. 2011 81(6): 1459-1473. </big> | ||
| + | [http://www.ncbi.nlm.nih.gov/pubmed/21815947 PubMed:21815947] | ||
<pubmed> 14976255, 3924737, 6436812,2422155,8419914, 1551827 </pubmed> | <pubmed> 14976255, 3924737, 6436812,2422155,8419914, 1551827 </pubmed> | ||
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 18:28, 19 November 2011
- Description: indole-3-glycerol-phosphate synthase
| Gene name | trpC |
| Synonyms | |
| Essential | no |
| Product | indole-3-glycerol-phosphate synthase |
| Function | biosynthesis of tryptophan |
| Metabolic function and regulation of this protein in SubtiPathways: Phe, Tyr, Trp | |
| MW, pI | 27 kDa, 5.535 |
| Gene length, protein length | 750 bp, 250 aa |
| Immediate neighbours | trpF, trpD |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 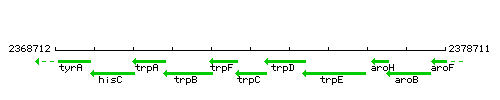
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU22660
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 1-(2-carboxyphenylamino)-1-deoxy-D-ribulose 5-phosphate = 1-C-(3-indolyl)-glycerol 3-phosphate + CO2 + H2O (according to Swiss-Prot)
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure: 1VC4 (from Thermus thermophilus, 36% identity, 49% similarity)
- UniProt: P03964
- KEGG entry: [3]
- E.C. number: 4.1.1.48
Additional information
Expression and regulation
- Operon:
- Regulatory mechanism:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Lehnik-Habrink M, Schaffer M, Mäder U, Diethmaier C, Herzberg C, Stülke J RNA processing in Bacillus subtilis: identification of targets of the essential RNase Y. Mol Microbiol. 2011 81(6): 1459-1473. PubMed:21815947