Difference between revisions of "SpeD"
| Line 80: | Line 80: | ||
* '''Structure:''' | * '''Structure:''' | ||
| − | * ''' | + | * '''UniProt:''' [http://www.uniprot.org/uniprot/O34426 O34426] |
* '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu:BSU29010] | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu:BSU29010] | ||
Revision as of 14:04, 20 July 2009
- Description: S-adenosylmethionine decarboxylase
| Gene name | speD |
| Synonyms | ytcF |
| Essential | no |
| Product | S-adenosylmethionine decarboxylase |
| Function | spermidine, polyamine biosynthesis |
| Metabolic function and regulation of this protein in SubtiPathways: Cys, Met & Sulfate assimilation, Central C-metabolism | |
| MW, pI | 13 kDa, 4.768 |
| Gene length, protein length | 384 bp, 128 aa |
| Immediate neighbours | ytcG, gapB |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 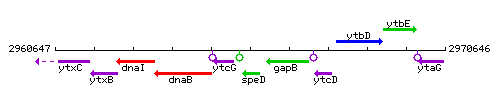
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU29010
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: S-adenosyl-L-methionine = (5-deoxy-5-adenosyl)(3-aminopropyl)-methylsulfonium salt + CO2 (according to Swiss-Prot)
- Protein family: Type 1 subfamily (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions:
- Localization:
Database entries
- Structure:
- UniProt: O34426
- KEGG entry: [3]
- E.C. number: 4.1.1.50
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
A Sekowska, J Y Coppée, J P Le Caer, I Martin-Verstraete, A Danchin
S-adenosylmethionine decarboxylase of Bacillus subtilis is closely related to archaebacterial counterparts.
Mol Microbiol: 2000, 36(5);1135-47
[PubMed:10844697]
[WorldCat.org]
[DOI]
(P p)