Difference between revisions of "MtnW"
| Line 29: | Line 29: | ||
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
|- | |- | ||
| − | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=mtnW_1427034_1428278_1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:mtnW_expression.png|500px]] | + | |colspan="2" |'''[http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=mtnW_1427034_1428278_1 Expression at a glance]'''   {{PubMed|22383849}}<br/>[[Image:mtnW_expression.png|500px|link=http://subtiwiki.uni-goettingen.de/apps/expression/expression.php?search=BSU13590]] |
|- | |- | ||
|} | |} | ||
Revision as of 13:06, 16 May 2013
- Description: 2,3-diketo-5-methylthiopentyl-1-phosphate enolase, Rubisco-like protein
| Gene name | mtnW |
| Synonyms | ykrW |
| Essential | no |
| Product | 2,3-diketo-5-methylthiopentyl-1-phosphate enolase |
| Function | methionine salvage |
| Gene expression levels in SubtiExpress: mtnW | |
| Metabolic function and regulation of this protein in SubtiPathways: Cys, Met & Sulfate assimilation | |
| MW, pI | 44 kDa, 5.284 |
| Gene length, protein length | 1242 bp, 414 aa |
| Immediate neighbours | mtnE, mtnX |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 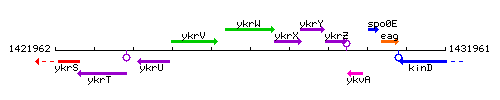
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed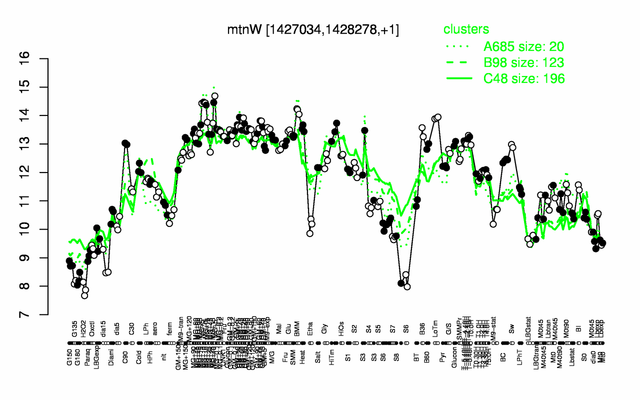
| |
Contents
Categories containing this gene/protein
biosynthesis/ acquisition of amino acids
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU13590
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: 5-(methylthio)-2,3-dioxopentyl phosphate = 2-hydroxy-5-(methylthio)-3-oxopent-1-enyl phosphate (according to Swiss-Prot)
- Protein family: Type IV subfamily (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- UniProt: O31666
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulatory mechanism: S-box: transcription termination/ antitermination, the S-box riboswitch binds S-adenosylmethionine resulting in termination PubMed
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Additional publications: PubMed
Haruka Tamura, Yohtaro Saito, Hiroki Ashida, Yasushi Kai, Tsuyoshi Inoue, Akiho Yokota, Hiroyoshi Matsumura
Structure of the apo decarbamylated form of 2,3-diketo-5-methylthiopentyl-1-phosphate enolase from Bacillus subtilis.
Acta Crystallogr D Biol Crystallogr: 2009, 65(Pt 9);942-51
[PubMed:19690372]
[WorldCat.org]
[DOI]
(I p)
Yohtaro Saito, Hiroki Ashida, Tomoko Sakiyama, Nicole Tandeau de Marsac, Antoine Danchin, Agnieszka Sekowska, Akiho Yokota
Structural and functional similarities between a ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO)-like protein from Bacillus subtilis and photosynthetic RuBisCO.
J Biol Chem: 2009, 284(19);13256-64
[PubMed:19279009]
[WorldCat.org]
[DOI]
(P p)
Haruka Tamura, Hiroki Ashida, Shogo Koga, Yohtaro Saito, Tomonori Yadani, Yasushi Kai, Tsuyoshi Inoue, Akiho Yokota, Hiroyoshi Matsumura
Crystallization and preliminary X-ray analysis of 2,3-diketo-5-methylthiopentyl-1-phosphate enolase from Bacillus subtilis.
Acta Crystallogr Sect F Struct Biol Cryst Commun: 2009, 65(Pt 2);147-50
[PubMed:19194007]
[WorldCat.org]
[DOI]
(I p)
Jerneja Tomsic, Brooke A McDaniel, Frank J Grundy, Tina M Henkin
Natural variability in S-adenosylmethionine (SAM)-dependent riboswitches: S-box elements in bacillus subtilis exhibit differential sensitivity to SAM In vivo and in vitro.
J Bacteriol: 2008, 190(3);823-33
[PubMed:18039762]
[WorldCat.org]
[DOI]
(I p)
Agnieszka Sekowska, Valérie Dénervaud, Hiroki Ashida, Karine Michoud, Dieter Haas, Akiho Yokota, Antoine Danchin
Bacterial variations on the methionine salvage pathway.
BMC Microbiol: 2004, 4;9
[PubMed:15102328]
[WorldCat.org]
[DOI]
(I e)
Hiroki Ashida, Yohtaro Saito, Chojiro Kojima, Kazuo Kobayashi, Naotake Ogasawara, Akiho Yokota
A functional link between RuBisCO-like protein of Bacillus and photosynthetic RuBisCO.
Science: 2003, 302(5643);286-90
[PubMed:14551435]
[WorldCat.org]
[DOI]
(I p)
Maumita Mandal, Benjamin Boese, Jeffrey E Barrick, Wade C Winkler, Ronald R Breaker
Riboswitches control fundamental biochemical pathways in Bacillus subtilis and other bacteria.
Cell: 2003, 113(5);577-86
[PubMed:12787499]
[WorldCat.org]
[DOI]
(P p)
Ulrike Mäder, Georg Homuth, Christian Scharf, Knut Büttner, Rüdiger Bode, Michael Hecker
Transcriptome and proteome analysis of Bacillus subtilis gene expression modulated by amino acid availability.
J Bacteriol: 2002, 184(15);4288-95
[PubMed:12107147]
[WorldCat.org]
[DOI]
(P p)
Agnieszka Sekowska, Antoine Danchin
The methionine salvage pathway in Bacillus subtilis.
BMC Microbiol: 2002, 2;8
[PubMed:12022921]
[WorldCat.org]
[DOI]
(I e)
Brooke A Murphy, Frank J Grundy, Tina M Henkin
Prediction of gene function in methylthioadenosine recycling from regulatory signals.
J Bacteriol: 2002, 184(8);2314-8
[PubMed:11914366]
[WorldCat.org]
[DOI]
(P p)
F J Grundy, T M Henkin
The S box regulon: a new global transcription termination control system for methionine and cysteine biosynthesis genes in gram-positive bacteria.
Mol Microbiol: 1998, 30(4);737-49
[PubMed:10094622]
[WorldCat.org]
[DOI]
(P p)