MreC
- Description: MreC is a cell shape determining protein and is associated with the MreB cytoskeleton in B. subtilis and other rod shaped bacteria.
| Gene name | mreC |
| Synonyms | |
| Essential | yes PubMed |
| Product | cell-shape determining protein |
| Function | cell-shape determation |
| MW, pI | 32 kDa, 6.248 |
| Gene length, protein length | 870 bp, 290 aa |
| Immediate neighbours | mreD, mreB |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 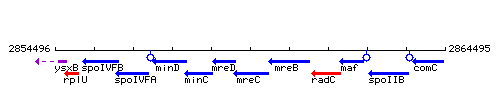
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU28020
Phenotypes of a mutant
mreC is essential under normal conditions PubMed. Depletion of MreC leads to a progressive increase in the width and a decrease in the length of the cell. This shape defect is consistent with a role for mreC in cell wall synthesis during elongation and has a similar phenotype to other genes with roles in elongation like rodA and the redundant gene pair pbpA and pbpH. Electron microscopy of cells depleted of MreC shows regions of the cell where a thick and irregular cell wall has accumulated PubMed. mreC can be deleted provided that 0.5 M sucrose and 20 mM Mg(2+) is provided in the media, mreC is therefore conditionally essentail. The phenotype of the mreC deletion in these conditions is one characterised by extreamly fat and bloated cells that tend to grow in clusters PubMed.
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
Function
MreC functions in cell wall synthesis by, together with the MreB cytoskeleton, localizing the cell wall synthetic machinery to the correct part of the cell. MreC therefore ensures that the cell wall is made in the correct way to maintain the proper shape of the cell.
MreC in other organisms
MreC has been studied in other organisms where it has been shown to be important in cell shape determination.
- Escherichia coli PubMed PubMed
- Caulobacter cresentus PubMed PubMed
- Rhodobacter spheroides PubMed
- Streptomyces coelicolor PubMed
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: None/ structural protein
- Protein family: mreC family (according to Swiss-Prot) COG1793
- Paralogous protein(s): None
Extended information on the protein
- Kinetic information:
- Domains: Intracellular N-terminus, transmembrane domain, Coiled coil domain and C-terminal beta-sheet domain.
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: cell membrane (according to Swiss-Prot), GFP-MreC localises to the cell membrane in a helical pattern PubMed.
Database entries
- Structure:
- UniProt: Q01466
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant: A non-polar inframe deletion strain named 3481 and a xylose dependent conditional mutant named 3461 is avaliable from the Errington lab PubMed.
- Expression vector: everest poker
- lacZ fusion:
- GFP fusion: A functional N-terminal GFP fusion has been made where the fusion protein is the only copy of the gene in the cell: strain 3417 PubMed.
- two-hybrid system:
- Antibody: antisera raised in rabit is avaliable from the Errington lab.
Labs working on this gene/protein
Jeff Errington, Newcastle University, UK homepage
Peter Graumann, Freiburg University, Germany homepage
Your additional remarks
mreC is an abbreviation of murein region e, gene C
References
Kathrin Schirner, Jeff Errington
Influence of heterologous MreB proteins on cell morphology of Bacillus subtilis.
Microbiology (Reading): 2009, 155(Pt 11);3611-3621
[PubMed:19643765]
[WorldCat.org]
[DOI]
(I p)
Emma J Hayhurst, Lekshmi Kailas, Jamie K Hobbs, Simon J Foster
Cell wall peptidoglycan architecture in Bacillus subtilis.
Proc Natl Acad Sci U S A: 2008, 105(38);14603-8
[PubMed:18784364]
[WorldCat.org]
[DOI]
(I p)
Alex Formstone, Rut Carballido-López, Philippe Noirot, Jeffery Errington, Dirk-Jan Scheffers
Localization and interactions of teichoic acid synthetic enzymes in Bacillus subtilis.
J Bacteriol: 2008, 190(5);1812-21
[PubMed:18156271]
[WorldCat.org]
[DOI]
(I p)
Felipe O Bendezú, Piet A J de Boer
Conditional lethality, division defects, membrane involution, and endocytosis in mre and mrd shape mutants of Escherichia coli.
J Bacteriol: 2008, 190(5);1792-811
[PubMed:17993535]
[WorldCat.org]
[DOI]
(I p)
Fusinita van den Ent, Mark Leaver, Felipe Bendezu, Jeff Errington, Piet de Boer, Jan Löwe
Dimeric structure of the cell shape protein MreC and its functional implications.
Mol Microbiol: 2006, 62(6);1631-42
[PubMed:17427287]
[WorldCat.org]
[DOI]
(P p)
Peter M Slovak, Steven L Porter, Judith P Armitage
Differential localization of Mre proteins with PBP2 in Rhodobacter sphaeroides.
J Bacteriol: 2006, 188(5);1691-700
[PubMed:16484180]
[WorldCat.org]
[DOI]
(P p)
Natalie A Dye, Zachary Pincus, Julie A Theriot, Lucy Shapiro, Zemer Gitai
Two independent spiral structures control cell shape in Caulobacter.
Proc Natl Acad Sci U S A: 2005, 102(51);18608-13
[PubMed:16344481]
[WorldCat.org]
[DOI]
(P p)
Arun V Divakaruni, Rachel R Ogorzalek Loo, Yongming Xie, Joseph A Loo, James W Gober
The cell-shape protein MreC interacts with extracytoplasmic proteins including cell wall assembly complexes in Caulobacter crescentus.
Proc Natl Acad Sci U S A: 2005, 102(51);18602-7
[PubMed:16344480]
[WorldCat.org]
[DOI]
(P p)
Mark Leaver, Jeff Errington
Roles for MreC and MreD proteins in helical growth of the cylindrical cell wall in Bacillus subtilis.
Mol Microbiol: 2005, 57(5);1196-209
[PubMed:16101995]
[WorldCat.org]
[DOI]
(P p)
Thomas Kruse, Jette Bork-Jensen, Kenn Gerdes
The morphogenetic MreBCD proteins of Escherichia coli form an essential membrane-bound complex.
Mol Microbiol: 2005, 55(1);78-89
[PubMed:15612918]
[WorldCat.org]
[DOI]
(P p)
Joong-Chul Lee, George C Stewart
Essential nature of the mreC determinant of Bacillus subtilis.
J Bacteriol: 2003, 185(15);4490-8
[PubMed:12867458]
[WorldCat.org]
[DOI]
(P p)
A Burger, K Sichler, G Kelemen, M Buttner, W Wohlleben
Identification and characterization of the mre gene region of Streptomyces coelicolor A3(2).
Mol Gen Genet: 2000, 263(6);1053-60
[PubMed:10954092]
[WorldCat.org]
[DOI]
(P p)
S Lee, C W Price
The minCD locus of Bacillus subtilis lacks the minE determinant that provides topological specificity to cell division.
Mol Microbiol: 1993, 7(4);601-10
[PubMed:8459776]
[WorldCat.org]
[DOI]
(P p)
P A Levin, P S Margolis, P Setlow, R Losick, D Sun
Identification of Bacillus subtilis genes for septum placement and shape determination.
J Bacteriol: 1992, 174(21);6717-28
[PubMed:1400224]
[WorldCat.org]
[DOI]
(P p)