Difference between revisions of "DhbF"
| Line 20: | Line 20: | ||
|style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 7134 bp, 2378 aa | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 7134 bp, 2378 aa | ||
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ | + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ybdZ]]'', ''[[dhbB]]'' |
|- | |- | ||
|colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB15186]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB15186]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' | ||
Revision as of 09:38, 5 December 2012
- Description: involved in 2,3-dihydroxybenzoate biosynthesis
| Gene name | dhbF |
| Synonyms | |
| Essential | no |
| Product | unknown |
| Function | biosynthesis of the siderophore bacillibactin |
| Gene expression levels in SubtiExpress: dhbF | |
| MW, pI | 263 kDa, 4.737 |
| Gene length, protein length | 7134 bp, 2378 aa |
| Immediate neighbours | ybdZ, dhbB |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 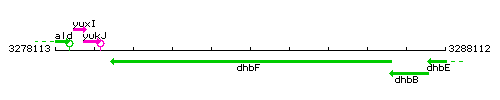
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed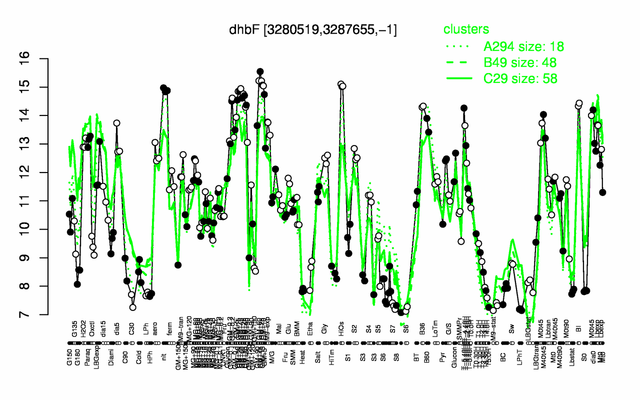
| |
Contents
Categories containing this gene/protein
acquisition of iron, iron metabolism, membrane proteins, phosphoproteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU31960
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: ATP-dependent AMP-binding enzyme family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification: phosphorylation on Ser-988 AND Ser-996 PubMed
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- membrane PubMed
Database entries
- Structure:
- UniProt: P45745
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Additional publications: PubMed
Lehnik-Habrink M, Schaffer M, Mäder U, Diethmaier C, Herzberg C, Stülke J RNA processing in Bacillus subtilis: identification of targets of the essential RNase Y. Mol Microbiol. 2011 81(6): 1459-1473. PubMed:21815947
Hannes Hahne, Susanne Wolff, Michael Hecker, Dörte Becher
From complementarity to comprehensiveness--targeting the membrane proteome of growing Bacillus subtilis by divergent approaches.
Proteomics: 2008, 8(19);4123-36
[PubMed:18763711]
[WorldCat.org]
[DOI]
(I p)
Boris Macek, Ivan Mijakovic, Jesper V Olsen, Florian Gnad, Chanchal Kumar, Peter R Jensen, Matthias Mann
The serine/threonine/tyrosine phosphoproteome of the model bacterium Bacillus subtilis.
Mol Cell Proteomics: 2007, 6(4);697-707
[PubMed:17218307]
[WorldCat.org]
[DOI]
(P p)
Noel Baichoo, Tao Wang, Rick Ye, John D Helmann
Global analysis of the Bacillus subtilis Fur regulon and the iron starvation stimulon.
Mol Microbiol: 2002, 45(6);1613-29
[PubMed:12354229]
[WorldCat.org]
[DOI]
(P p)
Tamara Hoffmann, Alexandra Schütz, Margot Brosius, Andrea Völker, Uwe Völker, Erhard Bremer
High-salinity-induced iron limitation in Bacillus subtilis.
J Bacteriol: 2002, 184(3);718-27
[PubMed:11790741]
[WorldCat.org]
[DOI]
(P p)
J J May, T M Wendrich, M A Marahiel
The dhb operon of Bacillus subtilis encodes the biosynthetic template for the catecholic siderophore 2,3-dihydroxybenzoate-glycine-threonine trimeric ester bacillibactin.
J Biol Chem: 2001, 276(10);7209-17
[PubMed:11112781]
[WorldCat.org]
[DOI]
(P p)
B M Rowland, T H Grossman, M S Osburne, H W Taber
Sequence and genetic organization of a Bacillus subtilis operon encoding 2,3-dihydroxybenzoate biosynthetic enzymes.
Gene: 1996, 178(1-2);119-23
[PubMed:8921902]
[WorldCat.org]
[DOI]
(P p)
B M Rowland, H W Taber
Duplicate isochorismate synthase genes of Bacillus subtilis: regulation and involvement in the biosyntheses of menaquinone and 2,3-dihydroxybenzoate.
J Bacteriol: 1996, 178(3);854-61
[PubMed:8550523]
[WorldCat.org]
[DOI]
(P p)