Difference between revisions of "CcpA"
(→Genes controlled by CcpA) |
(→Genes controlled by CcpA) |
||
| Line 66: | Line 66: | ||
* '''Activation by CcpA:''' ''[[pta]]'', ''[[ackA]]'', ''[[ilvB]]-[[ilvH]]-[[ilvC]]-[[leuA]]-[[leuB]]-[[leuC]]-[[leuD]]'' | * '''Activation by CcpA:''' ''[[pta]]'', ''[[ackA]]'', ''[[ilvB]]-[[ilvH]]-[[ilvC]]-[[leuA]]-[[leuB]]-[[leuC]]-[[leuD]]'' | ||
| − | * '''Repression by CcpA:''' ''[[amyE]]'', ''[[bglP]]-[[bglH]]'', ''[[bglS]]'', ''[[cccA]]'', ''[[levD]]-[[levE]]-[[levF]]-[[levG]]-[[sacC]]'', ''[[licB]]-[[licC]]-[[licA]]-[[licH]]'' | + | * '''Repression by CcpA:''' ''[[amyE]]'', ''[[bglP]]-[[bglH]]'', ''[[bglS]]'', ''[[cccA]]'', ''[[citZ]]-[[icd]]-[[mdh]]'', ''[[levD]]-[[levE]]-[[levF]]-[[levG]]-[[sacC]]'', ''[[licB]]-[[licC]]-[[licA]]-[[licH]]'' |
=== Extended information on the protein === | === Extended information on the protein === | ||
Revision as of 15:48, 9 February 2009
- Description: Carbon catabolite control protein A, involved in glucose regulation of many genes; represses catabolic genes and activates genes involved in excretion of excess carbon
| Gene name | ccpA |
| Synonyms | graR, alsA, amyR |
| Essential | no |
| Product | transcriptional regulator |
| Function | mediates carbon catabolite repression (CCR) |
| MW, pI | 36,8 kDa, 5.06 |
| Gene length, protein length | 1002 bp, 334 amino acids |
| Immediate neighbours | aroA, motP |
| Gene sequence (+200bp) | Protein sequence |
Genetic context 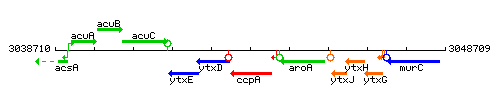
| |
Contents
The gene
Basic information
- Coordinates: 3043210 - 3044211
Phenotypes of a mutant
Loss of carbon catabolite repression. Loss of PTS-dependent sugar transport due to excessive phosphorylation of HPr by HprK. The mutant is unable to grow on a minimal medium with glucose and ammonium as the only sources of carbon and nitrogen, respectively.
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: transcriptional regulator of carbon catabolite repression (CCR)
- Protein family: LacI family
- Paralogous protein(s):
Genes controlled by CcpA
- Repression by CcpA: amyE, bglP-bglH, bglS, cccA, citZ-icd-mdh, levD-levE-levF-levG-sacC, licB-licC-licA-licH
Extended information on the protein
- Kinetic information:
- Domains:
- HTH lacI-type Domain (1 – 58)
- DNA binding Domain (6 – 25)
- Modification:
- Cofactor(s): HPr-Ser46-P, Crh-Ser-46-P
- Effectors of protein activity:glucose-6-phosphate, fructose-1,6-bisphosphate Pubmed
- Localization:
Database entries
- Swiss prot entry: [3]
- KEGG entry: [4]
Additional information
Expression and regulation
- Sigma factor:
- Regulation: constitutively expressed PubMed
- Additional information: there are about 3.000 molecules of CcpA per cell PubMed
Biological materials
- Mutant: QB5407 (spc), GP302 (erm), GP300 (an in frame deletion of ccpA), available in Stülke lab
- Expression vector: pGP643 (in pGP380, for SPINE, expression in Bacillus subtilis)
- lacZ fusion:
- GFP fusion:
Labs working on this gene/protein
Wolfgang Hillen, Erlangen University, Germany Homepage
Richard Brennan, Houston, Texas, USA Homepage
Milton H. Saier, University of California at San Diego, USA Homepage
Yasutaro Fujita, University of Fukuyama, Japan
Jörg Stülke, University of Göttingen, Germany Homepage
Your additional remarks
References
Reviews
- Henkin, T. M. (1996) The role of the CcpA transcriptional regulator in carbon metabolism in Bacillus subtilis. FEMS Microbiol Lett 135: 9-15. PubMed
- Warner, J. B. & Lolkema, J. S. CcpA-dependent carbon catabolite repression in bacteria. Microbiol. Mol. Biol. Rev. 67, 475-490 (2003). PubMed
General and physiological studies
- Henkin, T. M., Grundy, F. J., Nicholson, W. L. and Chambliss, G. H. (1991) Catabolite repression of -amylase gene expression in Bacillus subtilis involves a trans-acting gene product homologous to the Escherichia coli lacI and galR repressors. Mol. Microbiol. 5, 575-584. PubMed
- Faires, N., Tobisch, S., Bachem, S., Martin-Verstraete, I., Hecker, M., & Stülke, J. (1999) The catabolite control protein CcpA controls ammonium assimilation in Bacillus subtilis. J. Mol. Microbiol. Biotechnol. 1: 141-148. PubMed
- Ludwig, H., Rebhan, N., Blencke, H.-M., Merzbacher, M. & Stülke, J. (2002) Control of the glycolytic gapA operon by the catabolite control protein A in Bacillus subtilis: a novel mechanism of CcpA-mediated regulation. Mol. Microbiol. 45: 543-553. PubMed
- Miwa, Y., M. Saikawa, and Y. Fujita. 1994. Possible function and some properties of the CcpA protein of Bacillus subtilis. Microbiology 140:2567-2575. PubMed
- Singh, K. D., Schmalisch, M. H., Stülke, J. & Görke, B. (2008) Carbon catabolite repression in Bacillus subtilis: A quantitative analysis of repression exerted by different carbon sources. J. Bacteriol. 190: 7275-7284. PubMed
- Terahara et al. (2006) An intergenic stem-loop mutation in the Bacillus subtilis ccpA-motPS operon increases motPS transcription and the MotPS contribution to motility J Bacteriol. 188: 2701-2705. PubMed
- Wacker, I., Ludwig, H., Reif, I., Blencke, H.-M., Detsch, C. & Stülke, J. (2003) The regulatory link between carbon and nitrogen metabolism in Bacillus subtilis: regulation of the gltAB operon by the catabolite control protein CcpA. Microbiology 149: 3001-3009. PubMed
Global analyses (proteome, transcriptome)
- Blencke, H.-M., Homuth, G., Ludwig, H., Mäder, U., Hecker, M. & Stülke, J. (2003) Transcriptional profiling of gene expression in response to glucose in Bacillus subtilis: regulation of the central metabolic pathways. Metab. Engn. 5: 133-149. PubMed
- Moreno MS, Schneider BL, Maile RR, Weyler W, Saier Jr MH: Catabolite repression mediated by CcpA protein in Bacillus subtilis: novel modes of regulation revealed by whole-genome analysis. Mol Microbiol 2001, 39:1366-1381. PubMed
- Tobisch, S., Zühlke, D., Bernhardt, J., Stülke, J. & Hecker, M. (1999) Role of CcpA in regulation of the central pathways of carbon catabolism in Bacillus subtilis. J. Bacteriol. 181: 6996-7004. PubMed
- Yoshida, K.-I., Kobayashi, K., Miwa, Y., Kang, C.-M., Matsunaga, M., Yamaguchi, H., Tojo, S., Yamamoto, M., Nishi, R., Ogasawara, N., Nakayama, T. & Fujita, Y. (2001). Combined transcriptome and proteome analysis as a powerful approach to study genes under glucose repression in Bacillus subtilis. Nucl Acids Res 29, 6683-6692. PubMed
- Lulko, A. T., G. Buist, J. Kok, and O. P. Kuipers. 2007. Transcriptome analysis of temporal regulation of carbon metabolism by CcpA in Bacillus subtilis reveals additional target genes. J. Mol. Microbiol. Biotechnol. 12:82-95. PubMed
Repression of target genes by CcpA
- Belitsky BR, Sonenshein, AL: CcpA-dependent regulation of Bacillus subtilis glutamate dehydrogenase gene expression. J Bacteriol 2004, 186:3392-3398. PubMed
- Choi SK, Saier MH Jr: Regulation of sigL expression by the catabolite control protein CcpA involves a roadblock mechanism in Bacillus subtilis: potential connection between carbon and nitrogen metabolism. J Bacteriol 2005, 187:6856-6861. PubMed
- Darbon, E., Servant, P., Poncet, S., and Deutscher, J. (2002). Antitermination by GlpP, catabolite repression via CcpA and inducer exclusion triggered by P~GlpK dephosphorylation control Bacillus subtilis glpFK expression. Mol. Microbiol. 43, 1039-1052. PubMed
- Grundy, F. J., Turinski, A. J., and Henkin, T. M. (1994). Catabolite regulation of Bacillus subtilis acetate and acetoin utilization genes by CcpA. J. Bacteriol. 176, 4527-4533. PubMed
- Inacio, J. M. & de Sá-Nogueira, I. trans-Acting factors and cis-elements involved in glucose repression of arabinan degradation in Bacillus subtilis. J. Bacteriol. 189, 8371-8376 (2007). PubMed
- Kim HJ, Jourlin-Castelli C, Kim SI, Sonenshein AL (2002) Regulation of the Bacillus subtilis ccpC gene by CcpA and CcpC. Mol Microbiol 43:399-410 PubMed
- Kim HJ, Roux A, Sonenshein AL (2002) Direct and indirect roles of CcpA in regulation of Bacillus subtilis Krebs cycle genes. Mol Microbiol 45:179-190 PubMed
- Martin-Verstraete, I., Stülke, J., Klier, A. & Rapoport, G. (1995) Two different mechanisms mediate catabolite repression of the Bacillus subtilis levanase operon. J. Bacteriol. 177: 6919-6927. PubMed
Positive regulation of gene expression by CcpA
- Grundy FJ, Waters DA, Allen SH, Henkin TM (1993) Regulation of the Bacillus subtilis acetate kinase gene by CcpA. J Bacteriol 175:7348-7355 PubMed
- Ludwig, H., Meinken, C., Matin, A. & Stülke, J. (2002) Insufficient expression of the ilv-leu operon encoding enzymes of branched-chain amino acids biosynthesis limits growth of a Bacillus subtilis ccpA mutant. J. Bacteriol. 184: 5174-5178. PubMed
- Presecan-Siedel, E., Galinier, A., Longin, R., Deutscher, J., Danchin, A., Glaser, P. and Martin-Verstraete, I. (1999) The catabolite regulation of the pta gene as part of carbon flow pathways in Bacillus subtilis. J. Bacteriol. 181, 6889-6897. PubMed
- Shivers, R. P., and Sonenshein, A. L. (2005) Bacillus subtilis ilvB operon: an intersection of global regulons. Mol Microbiol 56: 1549-1559. PubMed
- Turinsky, A. J., Grundy, F. J., Kim, J. H., Chambliss, G. H., and Henkin, T. M. 1998. Transcriptional activation of the Bacillus subtilis ackA gene requires sequences upstream of the promoter. J. Bacteriol. 180: 5961-5967. PubMed
- Turinsky, A. J., Moir-Blais, T. R., Grundy, F. J., and Henkin, T. M. 2000. Bacillus subtilis ccpA gene mutants specifically defective in activation of acetoin synthesis. J. Bacteriol. 182:5611-5614. PubMed
Control of CcpA activity
- Deutscher, J., Küster, E., Bergstedt, U., Charrier, V., and Hillen, W. 1995. Protein kinase-dependent HPr/CcpA interaction links glycolytic activity to carbon catabolite repression in Gram-positive bacteria. Mol. Microbiol. 15: 1049-1053. PubMed
- Galinier A, Deutscher J, Martin-Verstraete I: Phosphorylation of either Crh or HPr mediates binding of CcpA to the Bacillus subtilis xyn cre and catabolite repression of the xyn operon. J Mol Biol 1999, 286:307-314. PubMed
- Jones, B. E., Dossonnet, V., Küster, E., Hillen, W., Deutscher, J. & Klevit, R. E. (1997). Binding of the catabolite repressor protein CcpA to its DNA target is regulated by phosphorylation of its corepressor HPr. J Biol Chem 272, 26530-26535. PubMed
- Aung-Hilbrich LM, Seidel G, Wagner A, Hillen W (2002) Quantification of the influence of HPrSer46P on CcpA-cre interaction. J Mol Biol 319:77-85. PubMed
- Kim JH, Voskuil MI, Chambliss GH (1998) NADP, corepressor for the Bacillus subtilis catabolite control protein CcpA. Proc Natl Acad Sci USA 95:9590-9595. PubMed
CcpA-DNA interaction
- Fujita, Y., Miwa, Y., Galinier, A. and Deutscher, J. (1995) Specific recognition of the Bacillus subtilis gnt cis-acting catabolite-responsive element by a protein complex formed between CcpA and seryl-phosphorylated HPr. Mol. Microbiol. 17, 953-960. PubMed
- Miwa, Y., Nakata, A., Ogiwara, A., Yamamota, M., and Fujita, Y. 2000. Evaluation and characterization of catabolite-responsive elements (cre) of Bacillus subtilis. Nucl. Acids Res. 28: 1206-1210. PubMed
- Seidel G, Diel M, Fuchsbauer N, Hillen W: Quantitative interdependence of coeffectors, CcpA and cre in carbon catabolite regulation of Bacillus subtilis. FEBS J 2005, 272:2566-2577. PubMed
- Kim JH, Guvener ZT, Cho JY, Chung KC, Chambliss GH (1995) Specificity of DNA binding activity of the Bacillus subtilis catabolite control protein CcpA. J Bacteriol 177: 5129-5134. PubMed
- Kim JH, Chambliss GH (1997) Contacts between Bacillus subtilis catabolite regulatory protein CcpA and amyO target site. Nucl Acids Res 25: 3490-3496. PubMed
Functional analysis of CcpA
- Küster, E., Hilbich, T., Dahl, M. and Hillen, W. (1999) Mutations in catabolite control protein CcpA separating growth effects from catabolite repression. J. Bacteriol. 181, 4125-4128. PubMed
- Küster-Schöck, E., Wagner, A., Völker, U., and Hillen, W. (1999) Mutations in catabolite control protein CcpA showing glucose-independent regulation in Bacillus megaterium. J Bacteriol 181: 7634-7638. PubMed
- Ludwig, H. & Stülke, J. (2001) The Bacillus subtilis catabolite control protein CcpA exerts all its regulatory functions by DNA binding. FEMS Microbiol. Lett. 203: 125-129. PubMed
- Kraus A, Hillen W. 1997. Analysis of ccpA mutations defective in carbon catabolite repression in Bacillus megaterium. FEMS Microbiol. Lett. 153:221-226. PubMed
- Kraus A, Küster E, Wagner A, Hoffmann K, Hillen W. 1998. Identification of a corepressor binding site in catabolite control protein CcpA. Mol. Microbiol. 30:955-963. PubMed
Structural analyses
- Schumacher, M. A. et al. Structural basis for allosteric control of the transcription regulator CcpA by the phosphoprotein HPr-Ser46-P. Cell 118, 731-741 (2004). PubMed
- Schumacher, M. A., Seidel, G., Hillen, W. & Brennan, R. G. Phosphoprotein Crh-Ser46-P displays altered binding to CcpA to effect carbon catabolite regulation. J. Biol. Chem. 281, 6793-6800 (2006). PubMed
- Schumacher, M. A., Seidel, G., Hillen, W. & Brennan, R. G. Structural mechanism for the fine-tuning of CcpA function by the small molecule effectors glucose 6-phosphate and fructose 1,6-bisphosphate. J. Mol. Biol. 368, 1042-1050 (2007). PubMed