Ugd
- Description: UDP-glucose dehydrogenase
| Gene name | ugd |
| Synonyms | ywqF |
| Essential | no |
| Product | UDP-glucose dehydrogenase |
| Function | unknown |
| Gene expression levels in SubtiExpress: ugd | |
| Interactions involving this protein in SubtInteract: Ugd | |
| MW, pI | 47 kDa, 5.03 |
| Gene length, protein length | 1320 bp, 440 aa |
| Immediate neighbours | ywqG, ptpZ |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed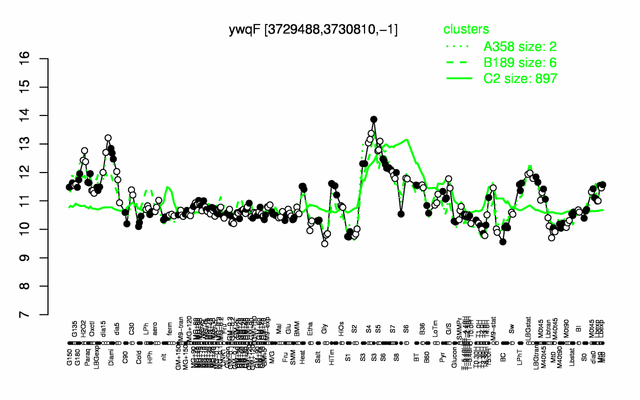
| |
Contents
Categories containing this gene/protein
phosphoproteins, sporulation proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU36230
Phenotypes of a mutant
Database entries
- BsubCyc: BSU36230
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: UDP-glucose + 2 NAD+ + H2O = UDP-glucuronate + 2 NADH (according to Swiss-Prot)
- Protein family: UDP-glucose/GDP-mannose dehydrogenase family (according to Swiss-Prot)
Extended information on the protein
- Kinetic information:
- Localization: cytoplasm (according to Swiss-Prot)
Database entries
- BsubCyc: BSU36230
- Structure:
- UniProt: P96718
- KEGG entry: [3]
- E.C. number: 1.1.1.22
Additional information
Expression and regulation
- Regulation:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Lei Shi, Boyang Ji, Lorena Kolar-Znika, Ana Boskovic, Fanny Jadeau, Christophe Combet, Christophe Grangeasse, Damjan Franjevic, Emmanuel Talla, Ivan Mijakovic
Evolution of bacterial protein-tyrosine kinases and their relaxed specificity toward substrates.
Genome Biol Evol: 2014, 6(4);800-17
[PubMed:24728941]
[WorldCat.org]
[DOI]
(I p)
Taryn B Kiley, Nicola R Stanley-Wall
Post-translational control of Bacillus subtilis biofilm formation mediated by tyrosine phosphorylation.
Mol Microbiol: 2010, 78(4);947-63
[PubMed:20815827]
[WorldCat.org]
[DOI]
(I p)
Dina Petranovic, Christophe Grangeasse, Boris Macek, Mohammad Abdillatef, Virginie Gueguen-Chaignon, Sylvie Nessler, Josef Deutscher, Ivan Mijakovic
Activation of Bacillus subtilis Ugd by the BY-kinase PtkA proceeds via phosphorylation of its residue tyrosine 70.
J Mol Microbiol Biotechnol: 2009, 17(2);83-9
[PubMed:19258708]
[WorldCat.org]
[DOI]
(I p)
Ivan Mijakovic, Lucia Musumeci, Lutz Tautz, Dina Petranovic, Robert A Edwards, Peter Ruhdal Jensen, Tomas Mustelin, Josef Deutscher, Nunzio Bottini
In vitro characterization of the Bacillus subtilis protein tyrosine phosphatase YwqE.
J Bacteriol: 2005, 187(10);3384-90
[PubMed:15866923]
[WorldCat.org]
[DOI]
(P p)
Ivan Mijakovic, Dina Petranovic, Josef Deutscher
How tyrosine phosphorylation affects the UDP-glucose dehydrogenase activity of Bacillus subtilis YwqF.
J Mol Microbiol Biotechnol: 2004, 8(1);19-25
[PubMed:15741737]
[WorldCat.org]
[DOI]
(P p)
Ivan Mijakovic, Sandrine Poncet, Grégory Boël, Alain Mazé, Sylvie Gillet, Emmanuel Jamet, Paulette Decottignies, Christophe Grangeasse, Patricia Doublet, Pierre Le Maréchal, Josef Deutscher
Transmembrane modulator-dependent bacterial tyrosine kinase activates UDP-glucose dehydrogenases.
EMBO J: 2003, 22(18);4709-18
[PubMed:12970183]
[WorldCat.org]
[DOI]
(P p)