Difference between revisions of "Sandbox"
| Line 1: | Line 1: | ||
| − | * '''Description:''' | + | * '''Description:''' Enzyme I, general (non sugar-specific) component of the PTS. Enzyme I transfers the phosphoryl group from phosphoenolpyruvate (PEP) to the phosphoryl carrier protein ([[PtsH |HPr]]) <br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
|- | |- | ||
|style="background:#ABCDEF;" align="center"|'''Gene name''' | |style="background:#ABCDEF;" align="center"|'''Gene name''' | ||
| − | |'' | + | |''ptsI'' |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' | + | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' '' |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Essential''' || no | + | |style="background:#ABCDEF;" align="center"| '''Essential''' || no |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Product''' || | + | |style="background:#ABCDEF;" align="center"| '''Product''' || phosphotransferase system (PTS) enzyme I |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || | + | |style="background:#ABCDEF;" align="center"|'''Function''' || PTS-dependent sugar transport |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || | + | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 62,9 kDa, 4.59 |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || | + | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 1710 bp, 570 amino acids |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ | + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ptsH]]'', ''[[splA]]'' |
|- | |- | ||
| − | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS: | + | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB13264]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' |
|- | |- | ||
| − | |colspan="2" | '''Genetic context''' <br/> [[Image: | + | |- |
| + | |- | ||
| + | |colspan="2" | '''Genetic context''' <br/> [[Image:ptsI_context.gif]] | ||
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
|- | |- | ||
| Line 29: | Line 31: | ||
__TOC__ | __TOC__ | ||
| − | <br/><br/><br/> | + | <br/><br/><br/><br/> |
=The gene= | =The gene= | ||
| Line 35: | Line 37: | ||
=== Basic information === | === Basic information === | ||
| − | * '''Locus tag:''' | + | * '''Locus tag:''' BSU13910 |
===Phenotypes of a mutant === | ===Phenotypes of a mutant === | ||
| Line 41: | Line 43: | ||
=== Database entries === | === Database entries === | ||
| − | * '''DBTBS entry:''' | + | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ptsGHI.html] |
| − | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+ | + | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG10201] |
=== Additional information=== | === Additional information=== | ||
| − | |||
=The protein= | =The protein= | ||
| Line 52: | Line 53: | ||
=== Basic information/ Evolution === | === Basic information/ Evolution === | ||
| − | * '''Catalyzed reaction/ biological activity:''' | + | * '''Catalyzed reaction/ biological activity:''' Phosphoenolpyruvate + protein L-histidine = pyruvate + protein N(pi)-phospho-L-histidine (according to Swiss-Prot) PEP-dependent autophosphorylation on His-189, transfer of the phosphoryl group to [[PtsH |HPr]] (His-15) |
| − | * '''Protein family:''' | + | * '''Protein family:''' PEP-utilizing enzyme family (according to Swiss-Prot) PEP-utilizing enzyme family |
* '''Paralogous protein(s):''' | * '''Paralogous protein(s):''' | ||
| Line 63: | Line 64: | ||
* '''Domains:''' | * '''Domains:''' | ||
| + | **HPr binding site (N-Terminal Domain) | ||
| + | **pyruvate binding site (C-Terminal Domain) | ||
| + | **pyrophosphate/phosphate carrier histidine (central Domain) | ||
| − | * '''Modification:''' | + | * '''Modification:''' transient autophosphorylation on His-189, in vivo also phosphorylated on Ser-34 or Ser-36 [http://www.ncbi.nlm.nih.gov/pubmed/17218307 PubMed] |
| − | * '''Cofactor(s):''' | + | * '''Cofactor(s):''' Magnesium |
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| Line 72: | Line 76: | ||
* '''Interactions:''' | * '''Interactions:''' | ||
| − | * '''Localization:''' | + | * '''Localization:''' cytoplasm (according to Swiss-Prot), Cytoplasm |
=== Database entries === | === Database entries === | ||
| Line 78: | Line 82: | ||
* '''Structure:''' | * '''Structure:''' | ||
| − | * '''Swiss prot entry:''' [http://www.uniprot.org/uniprot/ | + | * '''Swiss prot entry:''' [http://www.uniprot.org/uniprot/P08838 P08838] |
| − | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+ | + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+BSU13910] |
| − | * '''E.C. number:''' [http://www.expasy.org/enzyme/3. | + | * '''E.C. number:''' [http://www.expasy.org/enzyme/2.7.3.9 2.7.3.9] 2.7.3.9] |
=== Additional information=== | === Additional information=== | ||
| + | |||
=Expression and regulation= | =Expression and regulation= | ||
| − | * '''Operon:''' ''[[ | + | * '''Operon:''' |
| − | + | **''[[ptsG]]-[[ptsH]]-[[ptsI]]'' | |
| − | * '' | + | **''[[ptsH]]-[[ptsI]]'' |
| − | * '''Regulation:''' | + | * '''[[Sigma factor]]:''' [[SigA]] [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Abstract&list_uids=+11902727 PubMed] |
| + | * '''Regulation:''' expression activated by glucose (4.3 fold) [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed], induction by glucose (''[[ptsG]]''), constitutive (''[[ptsH]]'') | ||
| − | * '''Regulatory mechanism:''' | + | * '''Regulatory mechanism:''' ''[[ptsG]]'': transcriptional antitermination via the [[GlcT]]-dependent RNA-switch |
* '''Additional information:''' | * '''Additional information:''' | ||
| Line 100: | Line 106: | ||
=Biological materials = | =Biological materials = | ||
| − | * '''Mutant:''' | + | * '''Mutant:''' GP864 (ermC), available in [[Stülke]] lab |
| − | * '''Expression vector:''' | + | * '''Expression vector:''' pAG3 (His-tag), available in [[Galinier]] lab |
| − | + | ||
* '''lacZ fusion:''' | * '''lacZ fusion:''' | ||
* '''GFP fusion:''' | * '''GFP fusion:''' | ||
| − | |||
| − | |||
* '''Antibody:''' | * '''Antibody:''' | ||
=Labs working on this gene/protein= | =Labs working on this gene/protein= | ||
| + | |||
| + | [[Josef Deutscher]], Paris-Grignon, France | ||
| + | |||
| + | [[Stülke|Jörg Stülke]], University of Göttingen, Germany [http://wwwuser.gwdg.de/~genmibio/stuelke.html Homepage] | ||
=Your additional remarks= | =Your additional remarks= | ||
| Line 118: | Line 126: | ||
=References= | =References= | ||
| − | <pubmed> | + | <pubmed>12850135 17218307, </pubmed> |
| − | # | + | # Blencke et al. (2003) Transcriptional profiling of gene expression in response to glucose in ''Bacillus subtilis'': regulation of the central metabolic pathways. ''Metab Eng.'' '''5:''' 133-149 [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed] |
| − | # | + | # Frisby, D., and Zuber, P. 1994. Mutations in pts cause catabolite-resistant sporulation and altered regulation of spo0H in Bacillus subtilis. J. Bacteriol. 176: 2587-2595. [http://www.ncbi.nlm.nih.gov/sites/entrez/8169206 PubMed] |
| + | # Macek B, Mijakovic I, Olsen JV (2007) The serine/threonine/tyrosine phosphoproteome of the model bacterium Bacillus subtilis. ''Mol Cell Proteomics'' '''6(4):''' 697-707. [http://www.ncbi.nlm.nih.gov/sites/entrez/17218307 PubMed] | ||
Revision as of 13:30, 8 June 2009
- Description: Enzyme I, general (non sugar-specific) component of the PTS. Enzyme I transfers the phosphoryl group from phosphoenolpyruvate (PEP) to the phosphoryl carrier protein (HPr)
| Gene name | ptsI |
| Synonyms | |
| Essential | no |
| Product | phosphotransferase system (PTS) enzyme I |
| Function | PTS-dependent sugar transport |
| MW, pI | 62,9 kDa, 4.59 |
| Gene length, protein length | 1710 bp, 570 amino acids |
| Immediate neighbours | ptsH, splA |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 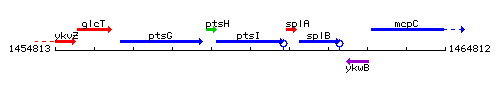
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU13910
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Phosphoenolpyruvate + protein L-histidine = pyruvate + protein N(pi)-phospho-L-histidine (according to Swiss-Prot) PEP-dependent autophosphorylation on His-189, transfer of the phosphoryl group to HPr (His-15)
- Protein family: PEP-utilizing enzyme family (according to Swiss-Prot) PEP-utilizing enzyme family
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- HPr binding site (N-Terminal Domain)
- pyruvate binding site (C-Terminal Domain)
- pyrophosphate/phosphate carrier histidine (central Domain)
- Modification: transient autophosphorylation on His-189, in vivo also phosphorylated on Ser-34 or Ser-36 PubMed
- Cofactor(s): Magnesium
- Effectors of protein activity:
- Interactions:
- Localization: cytoplasm (according to Swiss-Prot), Cytoplasm
Database entries
- Structure:
- Swiss prot entry: P08838
- KEGG entry: [3]
- E.C. number: 2.7.3.9 2.7.3.9]
Additional information
Expression and regulation
- Sigma factor: SigA PubMed
- Regulation: expression activated by glucose (4.3 fold) PubMed, induction by glucose (ptsG), constitutive (ptsH)
- Additional information:
Biological materials
- Mutant: GP864 (ermC), available in Stülke lab
- Expression vector: pAG3 (His-tag), available in Galinier lab
- lacZ fusion:
- GFP fusion:
- Antibody:
Labs working on this gene/protein
Josef Deutscher, Paris-Grignon, France
Jörg Stülke, University of Göttingen, Germany Homepage
Your additional remarks
References
Boris Macek, Ivan Mijakovic, Jesper V Olsen, Florian Gnad, Chanchal Kumar, Peter R Jensen, Matthias Mann
The serine/threonine/tyrosine phosphoproteome of the model bacterium Bacillus subtilis.
Mol Cell Proteomics: 2007, 6(4);697-707
[PubMed:17218307]
[WorldCat.org]
[DOI]
(P p)
Hans-Matti Blencke, Georg Homuth, Holger Ludwig, Ulrike Mäder, Michael Hecker, Jörg Stülke
Transcriptional profiling of gene expression in response to glucose in Bacillus subtilis: regulation of the central metabolic pathways.
Metab Eng: 2003, 5(2);133-49
[PubMed:12850135]
[WorldCat.org]
[DOI]
(P p)
- Blencke et al. (2003) Transcriptional profiling of gene expression in response to glucose in Bacillus subtilis: regulation of the central metabolic pathways. Metab Eng. 5: 133-149 PubMed
- Frisby, D., and Zuber, P. 1994. Mutations in pts cause catabolite-resistant sporulation and altered regulation of spo0H in Bacillus subtilis. J. Bacteriol. 176: 2587-2595. PubMed
- Macek B, Mijakovic I, Olsen JV (2007) The serine/threonine/tyrosine phosphoproteome of the model bacterium Bacillus subtilis. Mol Cell Proteomics 6(4): 697-707. PubMed