Difference between revisions of "Sandbox"
| Line 1: | Line 1: | ||
| − | * '''Description:''' | + | * '''Description:''' "Catabolite repression HPr-like protein", Cofactor of the [[CcpA]] transcription factor<br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
|- | |- | ||
|style="background:#ABCDEF;" align="center"|'''Gene name''' | |style="background:#ABCDEF;" align="center"|'''Gene name''' | ||
| − | |'' | + | |''crh'' |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' '' | + | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || ''yvcM'' |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Essential''' || no | + | |style="background:#ABCDEF;" align="center"| '''Essential''' || no |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Product''' || | + | |style="background:#ABCDEF;" align="center"| '''Product''' || catabolite repression HPr-like protein |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || | + | |style="background:#ABCDEF;" align="center"|'''Function''' || catabolite repression |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || | + | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 9,2 kDa, 4.70 |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || | + | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 255 bp, 85 amino acids |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ | + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[yvcL]]'', ''[[yvcN]]'' |
|- | |- | ||
| − | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS: | + | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB15479]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' |
|- | |- | ||
| − | |colspan="2" | '''Genetic context''' <br/> [[Image: | + | |colspan="2" | '''Genetic context''' <br/> [[Image:crh_context.gif]] |
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
|- | |- | ||
| Line 35: | Line 35: | ||
=== Basic information === | === Basic information === | ||
| − | * '''Locus tag:''' | + | * '''Locus tag:''' BSU34740 |
===Phenotypes of a mutant === | ===Phenotypes of a mutant === | ||
| Line 41: | Line 41: | ||
=== Database entries === | === Database entries === | ||
| − | * '''DBTBS entry:''' | + | * '''DBTBS entry:''' no entry |
| − | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+ | + | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG12403] |
=== Additional information=== | === Additional information=== | ||
| − | |||
=The protein= | =The protein= | ||
| Line 54: | Line 53: | ||
* '''Catalyzed reaction/ biological activity:''' | * '''Catalyzed reaction/ biological activity:''' | ||
| − | * '''Protein family:''' | + | * '''Protein family:''' HPr family (according to Swiss-Prot) [[PtsH]],HPr family |
| − | * '''Paralogous protein(s):''' | + | * '''Paralogous protein(s):''' [[PtsH |HPr]] |
=== Extended information on the protein === | === Extended information on the protein === | ||
| Line 62: | Line 61: | ||
* '''Kinetic information:''' | * '''Kinetic information:''' | ||
| − | * '''Domains:''' | + | * '''Domains:''' HPr domain (1–85) |
| − | * '''Modification:''' | + | * '''Modification:''' phosphorylation on Ser46 by [[HprK]] [http://www.ncbi.nlm.nih.gov/sites/entrez/9237995 PubMed] |
* '''Cofactor(s):''' | * '''Cofactor(s):''' | ||
| Line 70: | Line 69: | ||
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| − | * '''Interactions:''' [[ | + | * '''Interactions:''' Crh-[[GapA]] [http://www.ncbi.nlm.nih.gov/sites/entrez/17142398 PubMed], Crh-[[CcpA]] [http://www.ncbi.nlm.nih.gov/sites/entrez/12972249 PubMed], [[HprK]]-Crh [http://www.ncbi.nlm.nih.gov/sites/entrez/12009882 PubMed] |
* '''Localization:''' | * '''Localization:''' | ||
| Line 76: | Line 75: | ||
=== Database entries === | === Database entries === | ||
| − | * '''Structure:''' | + | * '''Structure:''' [http://www.rcsb.org/pdb/explore.do?structureId=2AK7 2AK7] (dimeric Crh-Ser46-P), [http://www.rcsb.org/pdb/explore.do?structureId=1ZVV 1ZVV] (CcpA-Crh-DNA complex), [http://www.rcsb.org/pdb/explore.do?structureId=2RLZ 2RLZ] (dimer), [http://www.rcsb.org/pdb/explore.do?structureId=1MU4 1MU4], [http://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?Dopt=s&uid=24634 NCBI], dimer [http://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?Dopt=s&uid=64943 NCBI], CcpA-Crh-DNA complex [http://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?Dopt=s&uid=52326 NCBI], dimeric phosphor-Crh [http://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?Dopt=s&uid=39567 NCBI] |
| + | * '''Swiss prot entry:''' [http://www.uniprot.org/uniprot/O06976 O06976] | ||
| − | * ''' | + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+BSU34740] |
| − | + | === Additional information=== | |
| − | |||
| − | |||
| − | + | Crh does not possess the phosphorylation site used for PTS phosphotransfer (His-15 in [[PtsH]]), it can only be phosphorylated on Ser-46 | |
=Expression and regulation= | =Expression and regulation= | ||
| − | * '''Operon:''' ''[[ | + | * '''Operon:''' ''[[yvcI]]-[[yvcJ]]-[[yvcK]]-[[yvcL]]-[[crh]]-[[yvcN]]'' [http://www.ncbi.nlm.nih.gov/sites/entrez/9237995 PubMed] |
| − | * ''' | + | * '''Sigma factor:''' [[SigA]] [http://www.ncbi.nlm.nih.gov/sites/entrez/16272399 PubMed] |
| − | * '''Regulation:''' | + | * '''Regulation:''' very weak stimuation of expression by citrate and succinate [http://www.ncbi.nlm.nih.gov/sites/entrez/16272399 PubMed] |
| − | * '''Regulatory mechanism:''' | + | * '''Regulatory mechanism:''' |
| − | * '''Additional information:''' | + | * '''Additional information:''' Crh is weakly expressed. This results in part from a poorly conserved ribosomal binding site of the mRNA. [http://www.ncbi.nlm.nih.gov/sites/entrez/15126459 PubMed] |
=Biological materials = | =Biological materials = | ||
| − | * '''Mutant:''' | + | * '''Mutant:''' GP860 (aphA3), QB7097 (spc), available in [[Stülke]] lab |
| − | * '''Expression vector:''' | + | * '''Expression vector:''' pGP41 (N-terminal Strep-tag, purification from ''B. subtilis'', for [[SPINE]], in [[pGP380]]), pGP734 (C-terminal Strep-tag, purification from ''B. subtilis'', for [[SPINE]], in [[pGP382]]), available in [[Stülke]] lab |
| − | + | ||
| − | * '''lacZ fusion:''' | + | * '''lacZ fusion:''' see ''[[yvcI]]'' |
* '''GFP fusion:''' | * '''GFP fusion:''' | ||
| − | * '''two-hybrid system:''' | + | * '''two-hybrid system:''' B. pertussis adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Boris Görke| Görke]] lab |
| − | * '''Antibody:''' | + | * '''Antibody:''' available in [[Stülke]] lab (not very good) |
=Labs working on this gene/protein= | =Labs working on this gene/protein= | ||
| + | |||
| + | [[Boris Görke]], University of Göttingen, Germany | ||
| + | [http://wwwuser.gwdg.de/~genmibio/goerke.html Homepage] | ||
| + | |||
| + | [[Anne Galinier]], University of Marseille, France | ||
| + | |||
| + | [[Wolfgang Hillen]], Erlangen University, Germany [http://www.biologie.uni-erlangen.de/mibi/index2.html Homepage] | ||
| + | |||
| + | [[Richard Brennan]], Houston, Texas, USA [http://www.mdanderson.org/departments/biochem/display.cfm?id=556ef368-6c81-4043-b74f350d41dd06cb&method=displayfull&pn=a8427ebd-d0ff-11d4-80fd00508b603a14 Homepage] | ||
=Your additional remarks= | =Your additional remarks= | ||
| Line 118: | Line 125: | ||
=References= | =References= | ||
| − | <pubmed> | + | <pubmed>12972249 9973552 9237995 16272399 15126459 10217795 17142398 16316990, </pubmed> |
| − | # M | + | # Juy M, Penin F, Favier A (2003) Dimerization of Crh by reversible 3D domain swapping induces structural adjustments to its monomeric homologue Hpr. ''J Mol Biol.'' '''332(4):'''767-76. [http://www.ncbi.nlm.nih.gov/sites/entrez/12972249 PubMed] |
| − | # | + | # Galinier A, Deutscher J, Martin-Verstraete I: Phosphorylation of either Crh or HPr mediates binding of CcpA to the ''Bacillus subtilis xyn cre'' and catabolite repression of the ''xyn'' operon. J Mol Biol 1999, 286:307-314. [http://www.ncbi.nlm.nih.gov/sites/entrez/9973552 PubMed] |
| + | # Galinier, A., Haiech, J., Kilhoffer, M.-C., Jaquinod, M., Stülke, J., Deutscher, J., & Martin-Verstraete, I. (1997) The ''Bacillus subtilis crh'' gene encodes a HPr-like protein involved in carbon catabolite repression. Proc. Natl. Acad. Sci. USA 94: 8439-8444. [http://www.ncbi.nlm.nih.gov/sites/entrez/9237995 PubMed] | ||
| + | # Görke et al. (2005) YvcK of ''Bacillus subtilis'' is required for a normal cell shape and for growth on Krebs cycle intermediates and substrates of the pentose phosphate pathway. Microbiology 151: 3777-3791. [http://www.ncbi.nlm.nih.gov/sites/entrez/16272399 PubMed] | ||
| + | # Görke, B., Fraysse, L. & Galinier, A. Drastic differences in Crh and HPr synthesis levels reflect their different impacts on catabolite repression in ''Bacillus subtilis''. J. Bacteriol. 186, 2992-2995 (2004). [http://www.ncbi.nlm.nih.gov/sites/entrez/15126459 PubMed] | ||
| + | # Martin-Verstraete, I., Deutscher, J., and Galinier, A. (1999) Phosphorylation of HPr and Crh by HprK, early steps in the catabolite repression signalling pathway for the ''Bacillus subtilis'' levanase operon. J Bacteriol 181: 2966-2969. [http://www.ncbi.nlm.nih.gov/sites/entrez/10217795 PubMed] | ||
| + | # Pompeo ''et al.'' (2007) Interaction of GapA with HPr and its homologue, Crh: Novel levels of regulation of a key step of glycolysis in ''Bacillus subtilis''? J Bacteriol 189, 1154-1157.[http://www.ncbi.nlm.nih.gov/sites/entrez/17142398 PubMed] | ||
| + | # Schumacher, M. A., Seidel, G., Hillen, W. & Brennan, R. G. Phosphoprotein Crh-Ser46-P displays altered binding to CcpA to effect carbon catabolite regulation. J. Biol. Chem. 281, 6793-6800 (2006). [http://www.ncbi.nlm.nih.gov/sites/entrez/16316990 PubMed] | ||
Revision as of 13:13, 8 June 2009
- Description: "Catabolite repression HPr-like protein", Cofactor of the CcpA transcription factor
| Gene name | crh |
| Synonyms | yvcM |
| Essential | no |
| Product | catabolite repression HPr-like protein |
| Function | catabolite repression |
| MW, pI | 9,2 kDa, 4.70 |
| Gene length, protein length | 255 bp, 85 amino acids |
| Immediate neighbours | yvcL, yvcN |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 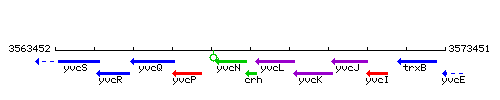
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU34740
Phenotypes of a mutant
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: HPr family (according to Swiss-Prot) PtsH,HPr family
- Paralogous protein(s): HPr
Extended information on the protein
- Kinetic information:
- Domains: HPr domain (1–85)
- Cofactor(s):
- Effectors of protein activity:
- Localization:
Database entries
- Structure: 2AK7 (dimeric Crh-Ser46-P), 1ZVV (CcpA-Crh-DNA complex), 2RLZ (dimer), 1MU4, NCBI, dimer NCBI, CcpA-Crh-DNA complex NCBI, dimeric phosphor-Crh NCBI
- Swiss prot entry: O06976
- KEGG entry: [2]
Additional information
Crh does not possess the phosphorylation site used for PTS phosphotransfer (His-15 in PtsH), it can only be phosphorylated on Ser-46
Expression and regulation
- Regulation: very weak stimuation of expression by citrate and succinate PubMed
- Regulatory mechanism:
- Additional information: Crh is weakly expressed. This results in part from a poorly conserved ribosomal binding site of the mRNA. PubMed
Biological materials
- Mutant: GP860 (aphA3), QB7097 (spc), available in Stülke lab
- Expression vector: pGP41 (N-terminal Strep-tag, purification from B. subtilis, for SPINE, in pGP380), pGP734 (C-terminal Strep-tag, purification from B. subtilis, for SPINE, in pGP382), available in Stülke lab
- lacZ fusion: see yvcI
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Görke lab
- Antibody: available in Stülke lab (not very good)
Labs working on this gene/protein
Boris Görke, University of Göttingen, Germany Homepage
Anne Galinier, University of Marseille, France
Wolfgang Hillen, Erlangen University, Germany Homepage
Richard Brennan, Houston, Texas, USA Homepage
Your additional remarks
References
Frédérique Pompeo, Jennifer Luciano, Anne Galinier
Interaction of GapA with HPr and its homologue, Crh: Novel levels of regulation of a key step of glycolysis in Bacillus subtilis?
J Bacteriol: 2007, 189(3);1154-7
[PubMed:17142398]
[WorldCat.org]
[DOI]
(P p)
Maria A Schumacher, Gerald Seidel, Wolfgang Hillen, Richard G Brennan
Phosphoprotein Crh-Ser46-P displays altered binding to CcpA to effect carbon catabolite regulation.
J Biol Chem: 2006, 281(10);6793-800
[PubMed:16316990]
[WorldCat.org]
[DOI]
(P p)
Boris Görke, Elodie Foulquier, Anne Galinier
YvcK of Bacillus subtilis is required for a normal cell shape and for growth on Krebs cycle intermediates and substrates of the pentose phosphate pathway.
Microbiology (Reading): 2005, 151(Pt 11);3777-3791
[PubMed:16272399]
[WorldCat.org]
[DOI]
(P p)
Boris Görke, Laetitia Fraysse, Anne Galinier
Drastic differences in Crh and HPr synthesis levels reflect their different impacts on catabolite repression in Bacillus subtilis.
J Bacteriol: 2004, 186(10);2992-5
[PubMed:15126459]
[WorldCat.org]
[DOI]
(P p)
Michel Juy, François Penin, Adrien Favier, Anne Galinier, Roland Montserret, Richard Haser, Josef Deutscher, Anja Böckmann
Dimerization of Crh by reversible 3D domain swapping induces structural adjustments to its monomeric homologue Hpr.
J Mol Biol: 2003, 332(4);767-76
[PubMed:12972249]
[WorldCat.org]
[DOI]
(P p)
I Martin-Verstraete, J Deutscher, A Galinier
Phosphorylation of HPr and Crh by HprK, early steps in the catabolite repression signalling pathway for the Bacillus subtilis levanase operon.
J Bacteriol: 1999, 181(9);2966-9
[PubMed:10217795]
[WorldCat.org]
[DOI]
(P p)
A Galinier, J Deutscher, I Martin-Verstraete
Phosphorylation of either crh or HPr mediates binding of CcpA to the bacillus subtilis xyn cre and catabolite repression of the xyn operon.
J Mol Biol: 1999, 286(2);307-14
[PubMed:9973552]
[WorldCat.org]
[DOI]
(P p)
A Galinier, J Haiech, M C Kilhoffer, M Jaquinod, J Stülke, J Deutscher, I Martin-Verstraete
The Bacillus subtilis crh gene encodes a HPr-like protein involved in carbon catabolite repression.
Proc Natl Acad Sci U S A: 1997, 94(16);8439-44
[PubMed:9237995]
[WorldCat.org]
[DOI]
(P p)
- Juy M, Penin F, Favier A (2003) Dimerization of Crh by reversible 3D domain swapping induces structural adjustments to its monomeric homologue Hpr. J Mol Biol. 332(4):767-76. PubMed
- Galinier A, Deutscher J, Martin-Verstraete I: Phosphorylation of either Crh or HPr mediates binding of CcpA to the Bacillus subtilis xyn cre and catabolite repression of the xyn operon. J Mol Biol 1999, 286:307-314. PubMed
- Galinier, A., Haiech, J., Kilhoffer, M.-C., Jaquinod, M., Stülke, J., Deutscher, J., & Martin-Verstraete, I. (1997) The Bacillus subtilis crh gene encodes a HPr-like protein involved in carbon catabolite repression. Proc. Natl. Acad. Sci. USA 94: 8439-8444. PubMed
- Görke et al. (2005) YvcK of Bacillus subtilis is required for a normal cell shape and for growth on Krebs cycle intermediates and substrates of the pentose phosphate pathway. Microbiology 151: 3777-3791. PubMed
- Görke, B., Fraysse, L. & Galinier, A. Drastic differences in Crh and HPr synthesis levels reflect their different impacts on catabolite repression in Bacillus subtilis. J. Bacteriol. 186, 2992-2995 (2004). PubMed
- Martin-Verstraete, I., Deutscher, J., and Galinier, A. (1999) Phosphorylation of HPr and Crh by HprK, early steps in the catabolite repression signalling pathway for the Bacillus subtilis levanase operon. J Bacteriol 181: 2966-2969. PubMed
- Pompeo et al. (2007) Interaction of GapA with HPr and its homologue, Crh: Novel levels of regulation of a key step of glycolysis in Bacillus subtilis? J Bacteriol 189, 1154-1157.PubMed
- Schumacher, M. A., Seidel, G., Hillen, W. & Brennan, R. G. Phosphoprotein Crh-Ser46-P displays altered binding to CcpA to effect carbon catabolite regulation. J. Biol. Chem. 281, 6793-6800 (2006). PubMed