Difference between revisions of "CotA"
(→References) |
(→Original publications) |
||
| Line 142: | Line 142: | ||
<pubmed>23202530</pubmed> | <pubmed>23202530</pubmed> | ||
== Original publications == | == Original publications == | ||
| − | <pubmed>15699190,3135411,11514528,1518043,2821284, 19933362, 20200715 20551082 20822511 21369750 22281748 23859715 22171814 22410485 15383836 24293734 24733162 </pubmed> | + | <pubmed>15699190,3135411,11514528,1518043,2821284, 19933362, 20200715 20551082 20822511 21369750 22281748 23859715 22171814 22410485 15383836 24293734 24733162 25259857 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 09:31, 29 September 2014
- Description: laccase, bilirubin oxidase, spore coat protein (outer)
| Gene name | cotA |
| Synonyms | pig |
| Essential | no |
| Product | laccase, bilirubin oxidase |
| Function | resistance of the spore |
| Gene expression levels in SubtiExpress: cotA | |
| MW, pI | 58 kDa, 5.89 |
| Gene length, protein length | 1539 bp, 513 aa |
| Immediate neighbours | yeaA, gabP |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 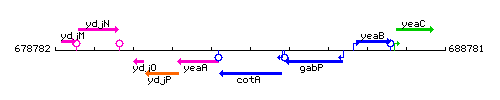
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed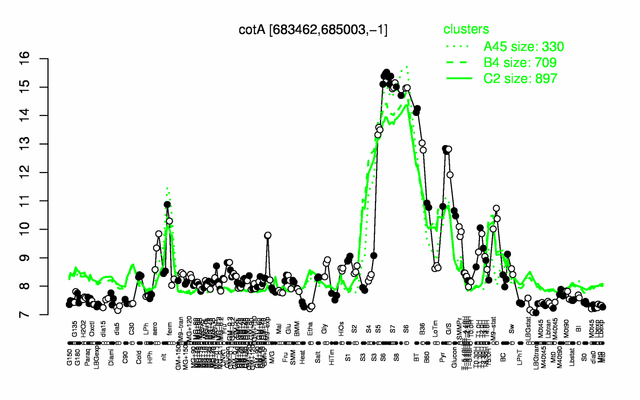
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
GerE regulon, SigK regulon, Efp-dependent proteins
The gene
Basic information
- Locus tag: BSU06300
Phenotypes of a mutant
Database entries
- BsubCyc: BSU06300
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Modification:
- Effectors of protein activity:
Database entries
- BsubCyc: BSU06300
- Structure: 2BHF (reduced form)
- UniProt: P07788
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: cotA (according to DBTBS)
- Additional information:
- translation is likely to require Efp due to the presence of several consecutive proline residues PubMed
Biological materials
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Peter T McKenney, Adam Driks, Patrick Eichenberger
The Bacillus subtilis endospore: assembly and functions of the multilayered coat.
Nat Rev Microbiol: 2013, 11(1);33-44
[PubMed:23202530]
[WorldCat.org]
[DOI]
(I p)
Original publications
Marco Plomp, Alicia Monroe Carroll, Peter Setlow, Alexander J Malkin
Architecture and assembly of the Bacillus subtilis spore coat.
PLoS One: 2014, 9(9);e108560
[PubMed:25259857]
[WorldCat.org]
[DOI]
(I e)
Thomas Beneyton, Faith Coldren, Jean-Christophe Baret, Andrew D Griffiths, Valérie Taly
CotA laccase: high-throughput manipulation and analysis of recombinant enzyme libraries expressed in E. coli using droplet-based microfluidics.
Analyst: 2014, 139(13);3314-23
[PubMed:24733162]
[WorldCat.org]
[DOI]
(I p)
Fatemeh Sheikhi, Mohammad Roayaei Ardakani, Naeimeh Enayatizamir, Susana Rodriguez-Couto
The Determination of Assay for Laccase of Bacillus subtilis WPI with Two Classes of Chemical Compounds as Substrates.
Indian J Microbiol: 2012, 52(4);701-7
[PubMed:24293734]
[WorldCat.org]
[DOI]
(P p)
Martiniano Bello, Jose Correa-Basurto, Enrique Rudiño-Piñera
Simulation of the cavity-binding site of three bacterial multicopper oxidases upon complex stabilization: interactional profile and electron transference pathways.
J Biomol Struct Dyn: 2014, 32(8);1303-17
[PubMed:23859715]
[WorldCat.org]
[DOI]
(I p)
Fabien Durand, Christian Hauge Kjaergaard, Emmanuel Suraniti, Sébastien Gounel, Ryan G Hadt, Edward I Solomon, Nicolas Mano
Bilirubin oxidase from Bacillus pumilus: a promising enzyme for the elaboration of efficient cathodes in biofuel cells.
Biosens Bioelectron: 2012, 35(1);140-146
[PubMed:22410485]
[WorldCat.org]
[DOI]
(I p)
Catarina S Silva, João M Damas, Zhenjia Chen, Vânia Brissos, Lígia O Martins, Cláudio M Soares, Peter F Lindley, Isabel Bento
The role of Asp116 in the reductive cleavage of dioxygen to water in CotA laccase: assistance during the proton-transfer mechanism.
Acta Crystallogr D Biol Crystallogr: 2012, 68(Pt 2);186-93
[PubMed:22281748]
[WorldCat.org]
[DOI]
(I p)
Peter T McKenney, Patrick Eichenberger
Dynamics of spore coat morphogenesis in Bacillus subtilis.
Mol Microbiol: 2012, 83(2);245-60
[PubMed:22171814]
[WorldCat.org]
[DOI]
(I p)
André T Fernandes, Manuela M Pereira, Catarina S Silva, Peter F Lindley, Isabel Bento, Eduardo Pinho Melo, Lígia O Martins
The removal of a disulfide bridge in CotA-laccase changes the slower motion dynamics involved in copper binding but has no effect on the thermodynamic stability.
J Biol Inorg Chem: 2011, 16(4);641-51
[PubMed:21369750]
[WorldCat.org]
[DOI]
(I p)
Isabel Bento, Catarina S Silva, Zhenjia Chen, Lígia O Martins, Peter F Lindley, Cláudio M Soares
Mechanisms underlying dioxygen reduction in laccases. Structural and modelling studies focusing on proton transfer.
BMC Struct Biol: 2010, 10;28
[PubMed:20822511]
[WorldCat.org]
[DOI]
(I e)
Nirupama Gupta, Edgardo T Farinas
Directed evolution of CotA laccase for increased substrate specificity using Bacillus subtilis spores.
Protein Eng Des Sel: 2010, 23(8);679-82
[PubMed:20551082]
[WorldCat.org]
[DOI]
(I p)
Zhenjia Chen, Paulo Durão, Catarina S Silva, Manuela M Pereira, Smilja Todorovic, Peter Hildebrandt, Isabel Bento, Peter F Lindley, Lígia O Martins
The role of Glu498 in the dioxygen reactivity of CotA-laccase from Bacillus subtilis.
Dalton Trans: 2010, 39(11);2875-82
[PubMed:20200715]
[WorldCat.org]
[DOI]
(I p)
Daisuke Imamura, Ritsuko Kuwana, Hiromu Takamatsu, Kazuhito Watabe
Localization of proteins to different layers and regions of Bacillus subtilis spore coats.
J Bacteriol: 2010, 192(2);518-24
[PubMed:19933362]
[WorldCat.org]
[DOI]
(I p)
Leif Steil, Mónica Serrano, Adriano O Henriques, Uwe Völker
Genome-wide analysis of temporally regulated and compartment-specific gene expression in sporulating cells of Bacillus subtilis.
Microbiology (Reading): 2005, 151(Pt 2);399-420
[PubMed:15699190]
[WorldCat.org]
[DOI]
(P p)
Patrick Eichenberger, Masaya Fujita, Shane T Jensen, Erin M Conlon, David Z Rudner, Stephanie T Wang, Caitlin Ferguson, Koki Haga, Tsutomu Sato, Jun S Liu, Richard Losick
The program of gene transcription for a single differentiating cell type during sporulation in Bacillus subtilis.
PLoS Biol: 2004, 2(10);e328
[PubMed:15383836]
[WorldCat.org]
[DOI]
(I p)
M F Hullo, I Moszer, A Danchin, I Martin-Verstraete
CotA of Bacillus subtilis is a copper-dependent laccase.
J Bacteriol: 2001, 183(18);5426-30
[PubMed:11514528]
[WorldCat.org]
[DOI]
(P p)
L Zheng, R Halberg, S Roels, H Ichikawa, L Kroos, R Losick
Sporulation regulatory protein GerE from Bacillus subtilis binds to and can activate or repress transcription from promoters for mother-cell-specific genes.
J Mol Biol: 1992, 226(4);1037-50
[PubMed:1518043]
[WorldCat.org]
[DOI]
(P p)
K Sandman, L Kroos, S Cutting, P Youngman, R Losick
Identification of the promoter for a spore coat protein gene in Bacillus subtilis and studies on the regulation of its induction at a late stage of sporulation.
J Mol Biol: 1988, 200(3);461-73
[PubMed:3135411]
[WorldCat.org]
[DOI]
(P p)
W Donovan, L B Zheng, K Sandman, R Losick
Genes encoding spore coat polypeptides from Bacillus subtilis.
J Mol Biol: 1987, 196(1);1-10
[PubMed:2821284]
[WorldCat.org]
[DOI]
(P p)