AccA
- Description: acetyl-CoA carboxylase (alpha subunit)
| Gene name | accA |
| Synonyms | |
| Essential | yes PubMed |
| Product | acetyl-CoA carboxylase (alpha subunit)) |
| Function | production of malonyl-CoA, the substrate for fatty acid biosynthesis |
| Gene expression levels in SubtiExpress: accA | |
| Interactions involving this protein in SubtInteract: AccA | |
| Metabolic function and regulation of this protein in SubtiPathways: accA | |
| MW, pI | 36 kDa, 6.087 |
| Gene length, protein length | 975 bp, 325 aa |
| Immediate neighbours | pfkA, accD |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 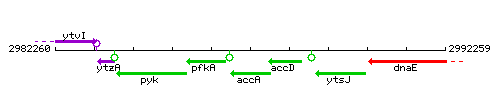
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed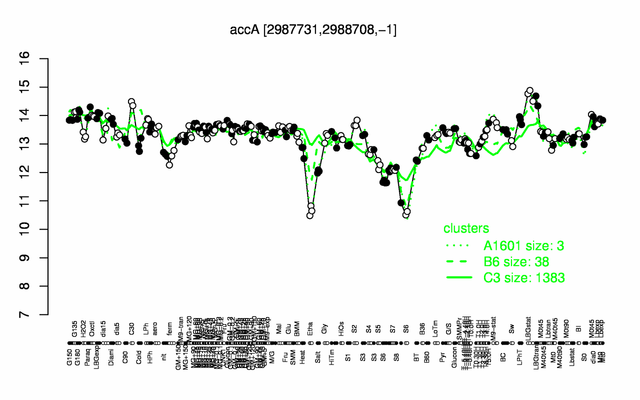
| |
Contents
Categories containing this gene/protein
biosynthesis of lipids, essential genes
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU29200
Phenotypes of a mutant
essential PubMed
Database entries
- BsubCyc: BSU29200
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATP + acetyl-CoA + HCO3- = ADP + phosphate + malonyl-CoA (according to Swiss-Prot)
- Protein family: accA family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: cytoplasm (according to Swiss-Prot), Membrane-proximal (Spotty) PubMed
Database entries
- BsubCyc: BSU29200
- UniProt: O34847
- KEGG entry: [3]
- E.C. number: 6.4.1.2
Additional information
Expression and regulation
- Sigma factor:
- Additional information:
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 1047 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 795 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 910 PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Reviews
Yasutaro Fujita, Hiroshi Matsuoka, Kazutake Hirooka
Regulation of fatty acid metabolism in bacteria.
Mol Microbiol: 2007, 66(4);829-39
[PubMed:17919287]
[WorldCat.org]
[DOI]
(P p)
Stephen W White, Jie Zheng, Yong-Mei Zhang, Rock
The structural biology of type II fatty acid biosynthesis.
Annu Rev Biochem: 2005, 74;791-831
[PubMed:15952903]
[WorldCat.org]
[DOI]
(P p)
Original Publications
Pierre Nicolas, Ulrike Mäder, Etienne Dervyn, Tatiana Rochat, Aurélie Leduc, Nathalie Pigeonneau, Elena Bidnenko, Elodie Marchadier, Mark Hoebeke, Stéphane Aymerich, Dörte Becher, Paola Bisicchia, Eric Botella, Olivier Delumeau, Geoff Doherty, Emma L Denham, Mark J Fogg, Vincent Fromion, Anne Goelzer, Annette Hansen, Elisabeth Härtig, Colin R Harwood, Georg Homuth, Hanne Jarmer, Matthieu Jules, Edda Klipp, Ludovic Le Chat, François Lecointe, Peter Lewis, Wolfram Liebermeister, Anika March, Ruben A T Mars, Priyanka Nannapaneni, David Noone, Susanne Pohl, Bernd Rinn, Frank Rügheimer, Praveen K Sappa, Franck Samson, Marc Schaffer, Benno Schwikowski, Leif Steil, Jörg Stülke, Thomas Wiegert, Kevin M Devine, Anthony J Wilkinson, Jan Maarten van Dijl, Michael Hecker, Uwe Völker, Philippe Bessières, Philippe Noirot
Condition-dependent transcriptome reveals high-level regulatory architecture in Bacillus subtilis.
Science: 2012, 335(6072);1103-6
[PubMed:22383849]
[WorldCat.org]
[DOI]
(I p)
Jean-Christophe Meile, Ling Juan Wu, S Dusko Ehrlich, Jeff Errington, Philippe Noirot
Systematic localisation of proteins fused to the green fluorescent protein in Bacillus subtilis: identification of new proteins at the DNA replication factory.
Proteomics: 2006, 6(7);2135-46
[PubMed:16479537]
[WorldCat.org]
[DOI]
(P p)
Patrick Bilder, Sandra Lightle, Graeme Bainbridge, Jeffrey Ohren, Barry Finzel, Fang Sun, Susan Holley, Loola Al-Kassim, Cindy Spessard, Michael Melnick, Marcia Newcomer, Grover L Waldrop
The structure of the carboxyltransferase component of acetyl-coA carboxylase reveals a zinc-binding motif unique to the bacterial enzyme.
Biochemistry: 2006, 45(6);1712-22
[PubMed:16460018]
[WorldCat.org]
[DOI]
(P p)
Christoph Freiberg, Nina A Brunner, Guido Schiffer, Thomas Lampe, Jens Pohlmann, Michael Brands, Martin Raabe, Dieter Häbich, Karl Ziegelbauer
Identification and characterization of the first class of potent bacterial acetyl-CoA carboxylase inhibitors with antibacterial activity.
J Biol Chem: 2004, 279(25);26066-73
[PubMed:15066985]
[WorldCat.org]
[DOI]
(P p)
Virginie Molle, Masaya Fujita, Shane T Jensen, Patrick Eichenberger, José E González-Pastor, Jun S Liu, Richard Losick
The Spo0A regulon of Bacillus subtilis.
Mol Microbiol: 2003, 50(5);1683-701
[PubMed:14651647]
[WorldCat.org]
[DOI]
(P p)
Hailong Zhang, Zhiru Yang, Yang Shen, Liang Tong
Crystal structure of the carboxyltransferase domain of acetyl-coenzyme A carboxylase.
Science: 2003, 299(5615);2064-7
[PubMed:12663926]
[WorldCat.org]
[DOI]
(I p)