AprE
- Description: extracellular alkaline serine protease (subtilisin E)
| Gene name | aprE |
| Synonyms | sprE |
| Essential | no |
| Product | extracellular alkaline serine protease (subtilisin E)) |
| Function | protein degradation |
| Interactions involving this protein in SubtInteract: AprE | |
| MW, pI | 39 kDa, 9.342 |
| Gene length, protein length | 1143 bp, 381 aa |
| Immediate neighbours | yhfN, yhfO |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 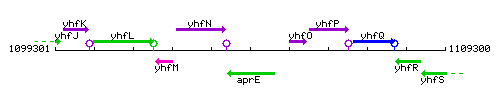
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
utilization of nitrogen sources other than amino acids
This gene is a member of the following regulons
AbrB regulon, ScoC regulon, SinR regulon
The gene
Basic information
- Locus tag: BSU10300
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: Hydrolysis of proteins with broad specificity for peptide bonds, and a preference for a large uncharged residue in P1 (according to Swiss-Prot)
- Protein family: peptidase S8 family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: secreted (according to Swiss-Prot), extracellular (signal peptide) PubMed
Database entries
- Structure: 1SBC
- UniProt: P04189
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: aprE (according to DBTBS)
- Regulatory mechanism:
- Additional information: the mRNA is extremely stable (more than 25 min) PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
- Additional information: The pAPNC213 vector published by Morimoto et al. (2002) is an insertion plasmid for IPTG inducible constructs that can be integrated into the chromosomal aprE locus by replacing the aprE open reading frame via double homologous recombination. PubMed
Labs working on this gene/protein
Your additional remarks
References
Reviews
Massimiliano Marvasi, Pieter T Visscher, Lilliam Casillas Martinez
Exopolymeric substances (EPS) from Bacillus subtilis: polymers and genes encoding their synthesis.
FEMS Microbiol Lett: 2010, 313(1);1-9
[PubMed:20735481]
[WorldCat.org]
[DOI]
(I p)
Original publications
Weihua Gao, Sen Dai, Quanli Liu, Haijin Xu, Mingqiang Qiao
CitB mutation increases the alkaline protease productivity in Bacillus subtilis.
J Gen Appl Microbiol: 2010, 56(5);403-7
[PubMed:21099137]
[WorldCat.org]
[DOI]
(P p)
Eliel R Romero-García, Alfredo Téllez-Valencia, María F Trujillo, José G Sampedro, Hugo Nájera, Arturo Rojo-Domínguez, Jesús García-Soto, Mario Pedraza-Reyes
Engineering and directed evolution of a Ca2+ binding site A-deficient AprE mutant reveal an essential contribution of the loop Leu75-Leu82 to enzyme activity.
J Biomed Biotechnol: 2009, 2009;201075
[PubMed:19710937]
[WorldCat.org]
[DOI]
(I p)
Sadanobu Abe, Ayako Yasumura, Teruo Tanaka
Regulation of Bacillus subtilis aprE expression by glnA through inhibition of scoC and sigma(D)-dependent degR expression.
J Bacteriol: 2009, 191(9);3050-8
[PubMed:19251843]
[WorldCat.org]
[DOI]
(I p)
Birgit Voigt, Haike Antelmann, Dirk Albrecht, Armin Ehrenreich, Karl-Heinz Maurer, Stefan Evers, Gerhard Gottschalk, Jan Maarten van Dijl, Thomas Schweder, Michael Hecker
Cell physiology and protein secretion of Bacillus licheniformis compared to Bacillus subtilis.
J Mol Microbiol Biotechnol: 2009, 16(1-2);53-68
[PubMed:18957862]
[WorldCat.org]
[DOI]
(I p)
Jan-Willem Veening, Oleg A Igoshin, Robyn T Eijlander, Reindert Nijland, Leendert W Hamoen, Oscar P Kuipers
Transient heterogeneity in extracellular protease production by Bacillus subtilis.
Mol Syst Biol: 2008, 4;184
[PubMed:18414485]
[WorldCat.org]
[DOI]
(I p)
Kana Shimane, Mitsuo Ogura
Mutational analysis of the helix-turn-helix region of Bacillus subtilis response regulator DegU, and identification of cis-acting sequences for DegU in the aprE and comK promoters.
J Biochem: 2004, 136(3);387-97
[PubMed:15598897]
[WorldCat.org]
[DOI]
(P p)
Mitsuo Ogura, Atsushi Matsuzawa, Hirofumi Yoshikawa, Teruo Tanaka
Bacillus subtilis SalA (YbaL) negatively regulates expression of scoC, which encodes the repressor for the alkaline exoprotease gene, aprE.
J Bacteriol: 2004, 186(10);3056-64
[PubMed:15126467]
[WorldCat.org]
[DOI]
(P p)
Atsuhiro Arai, Eiji Kawachi, Michihiro Hata, Mitsuo Ogura, Teruo Tanaka
Inhibition of Bacillus subtilis aprE expression by lincomycin at the posttranscriptional level through inhibition of ppGpp synthesis.
J Biochem: 2003, 134(5);691-7
[PubMed:14688235]
[WorldCat.org]
[DOI]
(P p)
G Hambraeus, C von Wachenfeldt, L Hederstedt
Genome-wide survey of mRNA half-lives in Bacillus subtilis identifies extremely stable mRNAs.
Mol Genet Genomics: 2003, 269(5);706-14
[PubMed:12884008]
[WorldCat.org]
[DOI]
(P p)
Gustav Hambraeus, Kaisa Karhumaa, Blanka Rutberg
A 5' stem-loop and ribosome binding but not translation are important for the stability of Bacillus subtilis aprE leader mRNA.
Microbiology (Reading): 2002, 148(Pt 6);1795-1803
[PubMed:12055299]
[WorldCat.org]
[DOI]
(P p)
T Urano, H Ihara, K Umemura, Y Suzuki, M Oike, S Akita, Y Tsukamoto, I Suzuki, A Takada
The profibrinolytic enzyme subtilisin NAT purified from Bacillus subtilis Cleaves and inactivates plasminogen activator inhibitor type 1.
J Biol Chem: 2001, 276(27);24690-6
[PubMed:11325965]
[WorldCat.org]
[DOI]
(P p)
Gustav Hambraeus, Martin Persson, Blanka Rutberg
The aprE leader is a determinant of extreme mRNA stability in Bacillus subtilis.
Microbiology (Reading): 2000, 146 Pt 12;3051-3059
[PubMed:11101663]
[WorldCat.org]
[DOI]
(P p)
P T Kallio, J E Fagelson, J A Hoch, M A Strauch
The transition state regulator Hpr of Bacillus subtilis is a DNA-binding protein.
J Biol Chem: 1991, 266(20);13411-7
[PubMed:1906467]
[WorldCat.org]
(P p)
N K Gaur, J Oppenheim, I Smith
The Bacillus subtilis sin gene, a regulator of alternate developmental processes, codes for a DNA-binding protein.
J Bacteriol: 1991, 173(2);678-86
[PubMed:1898931]
[WorldCat.org]
[DOI]
(P p)
M A Strauch, G B Spiegelman, M Perego, W C Johnson, D Burbulys, J A Hoch
The transition state transcription regulator abrB of Bacillus subtilis is a DNA binding protein.
EMBO J: 1989, 8(5);1615-21
[PubMed:2504584]
[WorldCat.org]
[DOI]
(P p)
M Perego, J A Hoch
Sequence analysis and regulation of the hpr locus, a regulatory gene for protease production and sporulation in Bacillus subtilis.
J Bacteriol: 1988, 170(6);2560-7
[PubMed:3131303]
[WorldCat.org]
[DOI]
(P p)
D J Henner, E Ferrari, M Perego, J A Hoch
Location of the targets of the hpr-97, sacU32(Hy), and sacQ36(Hy) mutations in upstream regions of the subtilisin promoter.
J Bacteriol: 1988, 170(1);296-300
[PubMed:2447063]
[WorldCat.org]
[DOI]
(P p)
E Ferrari, D J Henner, M Perego, J A Hoch
Transcription of Bacillus subtilis subtilisin and expression of subtilisin in sporulation mutants.
J Bacteriol: 1988, 170(1);289-95
[PubMed:2447062]
[WorldCat.org]
[DOI]
(P p)
E Ferrari, S M Howard, J A Hoch
Effect of stage 0 sporulation mutations on subtilisin expression.
J Bacteriol: 1986, 166(1);173-9
[PubMed:3082852]
[WorldCat.org]
[DOI]
(P p)
S L Wong, C W Price, D S Goldfarb, R H Doi
The subtilisin E gene of Bacillus subtilis is transcribed from a sigma 37 promoter in vivo.
Proc Natl Acad Sci U S A: 1984, 81(4);1184-8
[PubMed:6322190]
[WorldCat.org]
[DOI]
(P p)