Difference between revisions of "PdxS"
| Line 130: | Line 130: | ||
** number of protein molecules per cell (minimal medium with glucose and ammonium): 6001 {{PubMed|24696501}} | ** number of protein molecules per cell (minimal medium with glucose and ammonium): 6001 {{PubMed|24696501}} | ||
** number of protein molecules per cell (complex medium with amino acids, without glucose): 16290 {{PubMed|24696501}} | ** number of protein molecules per cell (complex medium with amino acids, without glucose): 16290 {{PubMed|24696501}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 7634 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 4624 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 7523 {{PubMed|21395229}} | ||
=Biological materials = | =Biological materials = | ||
| − | |||
* '''Mutant:''' | * '''Mutant:''' | ||
Revision as of 14:16, 17 April 2014
- Description: pyridoxal-5'-phosphate synthase (synthase domain)
| Gene name | pdxS |
| Synonyms | yaaD |
| Essential | no |
| Product | pyridoxal-5'-phosphate synthase (synthase domain) |
| Function | pyridoxal-5'-phosphate biosynthesis |
| Gene expression levels in SubtiExpress: pdxS | |
| Interactions involving this protein in SubtInteract: PdxS | |
| Metabolic function and regulation of this protein in SubtiPathways: PdxS | |
| MW, pI | 31 kDa, 5.085 |
| Gene length, protein length | 882 bp, 294 aa |
| Immediate neighbours | dacA, pdxT |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 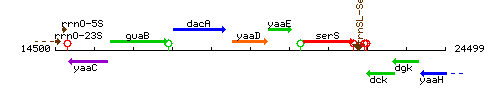
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed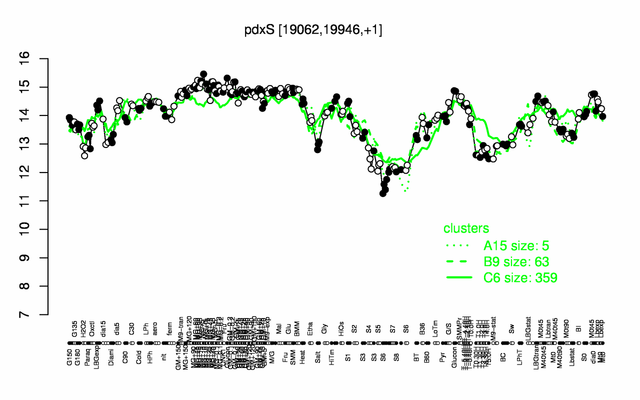
| |
Contents
Categories containing this gene/protein
biosynthesis of cofactors, phosphoproteins, most abundant proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU00110
Phenotypes of a mutant
auxotrophic for pyridoxal 5'-phosphate PubMed
Database entries
- BsubCyc: BSU00110
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: pdxS/SNZ family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Effectors of protein activity:
Database entries
- BsubCyc: BSU00110
- UniProt: P37527
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
- belongs to the 100 most abundant proteins PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium): 6001 PubMed
- number of protein molecules per cell (complex medium with amino acids, without glucose): 16290 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 7634 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 4624 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 7523 PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Alexander K W Elsholz, Kürsad Turgay, Stephan Michalik, Bernd Hessling, Katrin Gronau, Dan Oertel, Ulrike Mäder, Jörg Bernhardt, Dörte Becher, Michael Hecker, Ulf Gerth
Global impact of protein arginine phosphorylation on the physiology of Bacillus subtilis.
Proc Natl Acad Sci U S A: 2012, 109(19);7451-6
[PubMed:22517742]
[WorldCat.org]
[DOI]
(I p)
Silvia Wallner, Martina Neuwirth, Karlheinz Flicker, Ivo Tews, Peter Macheroux
Dissection of contributions from invariant amino acids to complex formation and catalysis in the heteromeric pyridoxal 5-phosphate synthase complex from Bacillus subtilis.
Biochemistry: 2009, 48(9);1928-35
[PubMed:19152323]
[WorldCat.org]
[DOI]
(I p)
Christine Eymann, Dörte Becher, Jörg Bernhardt, Katrin Gronau, Anja Klutzny, Michael Hecker
Dynamics of protein phosphorylation on Ser/Thr/Tyr in Bacillus subtilis.
Proteomics: 2007, 7(19);3509-26
[PubMed:17726680]
[WorldCat.org]
[DOI]
(P p)
Marco Strohmeier, Thomas Raschle, Jacek Mazurkiewicz, Karsten Rippe, Irmgard Sinning, Teresa B Fitzpatrick, Ivo Tews
Structure of a bacterial pyridoxal 5'-phosphate synthase complex.
Proc Natl Acad Sci U S A: 2006, 103(51);19284-9
[PubMed:17159152]
[WorldCat.org]
[DOI]
(P p)
Alain Lévine, Françoise Vannier, Cédric Absalon, Lauriane Kuhn, Peter Jackson, Elaine Scrivener, Valérie Labas, Joëlle Vinh, Patrick Courtney, Jérôme Garin, Simone J Séror
Analysis of the dynamic Bacillus subtilis Ser/Thr/Tyr phosphoproteome implicated in a wide variety of cellular processes.
Proteomics: 2006, 6(7);2157-73
[PubMed:16493705]
[WorldCat.org]
[DOI]
(P p)
Thomas Raschle, Nikolaus Amrhein, Teresa B Fitzpatrick
On the two components of pyridoxal 5'-phosphate synthase from Bacillus subtilis.
J Biol Chem: 2005, 280(37);32291-300
[PubMed:16030023]
[WorldCat.org]
[DOI]
(P p)
Jianghai Zhu, John W Burgner, Etti Harms, Boris R Belitsky, Janet L Smith
A new arrangement of (beta/alpha)8 barrels in the synthase subunit of PLP synthase.
J Biol Chem: 2005, 280(30);27914-23
[PubMed:15911615]
[WorldCat.org]
[DOI]
(P p)
Christine Eymann, Annette Dreisbach, Dirk Albrecht, Jörg Bernhardt, Dörte Becher, Sandy Gentner, Le Thi Tam, Knut Büttner, Gerrit Buurman, Christian Scharf, Simone Venz, Uwe Völker, Michael Hecker
A comprehensive proteome map of growing Bacillus subtilis cells.
Proteomics: 2004, 4(10);2849-76
[PubMed:15378759]
[WorldCat.org]
[DOI]
(P p)
Boris R Belitsky
Physical and enzymological interaction of Bacillus subtilis proteins required for de novo pyridoxal 5'-phosphate biosynthesis.
J Bacteriol: 2004, 186(4);1191-6
[PubMed:14762015]
[WorldCat.org]
[DOI]
(P p)
Virginie Molle, Masaya Fujita, Shane T Jensen, Patrick Eichenberger, José E González-Pastor, Jun S Liu, Richard Losick
The Spo0A regulon of Bacillus subtilis.
Mol Microbiol: 2003, 50(5);1683-701
[PubMed:14651647]
[WorldCat.org]
[DOI]
(P p)