Difference between revisions of "DhbE"
| Line 126: | Line 126: | ||
** number of protein molecules per cell (minimal medium with glucose and ammonium): 4899 {{PubMed|24696501}} | ** number of protein molecules per cell (minimal medium with glucose and ammonium): 4899 {{PubMed|24696501}} | ||
** number of protein molecules per cell (complex medium with amino acids, without glucose): 1656 {{PubMed|24696501}} | ** number of protein molecules per cell (complex medium with amino acids, without glucose): 1656 {{PubMed|24696501}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 10561 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 4162 {{PubMed|21395229}} | ||
| + | ** number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 3499 {{PubMed|21395229}} | ||
=Biological materials = | =Biological materials = | ||
| − | |||
* '''Mutant:''' | * '''Mutant:''' | ||
Revision as of 14:05, 17 April 2014
- Description: 2,3-dihydroxybenzoate-AMP ligase (enterobactin synthetase component E)
| Gene name | dhbE |
| Synonyms | entE |
| Essential | no |
| Product | 2,3-dihydroxybenzoate-AMP ligase (enterobactin synthetase component E) |
| Function | biosynthesis of the siderophore bacillibactin |
| Gene expression levels in SubtiExpress: dhbE | |
| Metabolic function and regulation of this protein in SubtiPathways: DhbE | |
| MW, pI | 59 kDa, 5.684 |
| Gene length, protein length | 1617 bp, 539 aa |
| Immediate neighbours | dhbB, dhbC |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 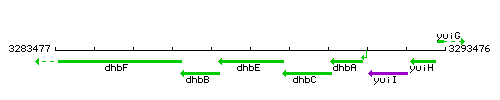
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed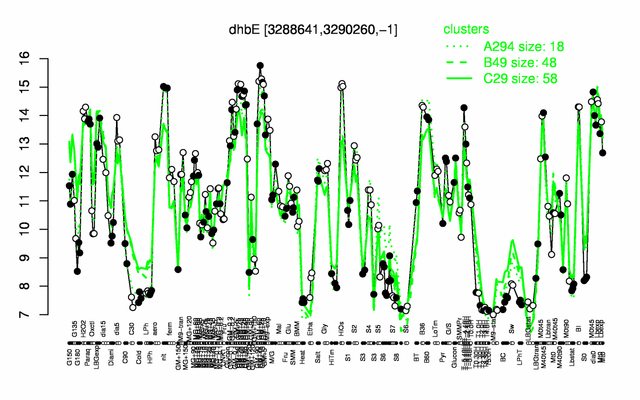
| |
Contents
Categories containing this gene/protein
acquisition of iron, iron metabolism
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU31980
Phenotypes of a mutant
Database entries
- BsubCyc: BSU31980
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: ATP-dependent AMP-binding enzyme family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- cytoplasm (according to Swiss-Prot)
Database entries
- BsubCyc: BSU31980
- UniProt: P40871
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
- the amount of the mRNA is substantially decreased upon depletion of RNase Y PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium): 4899 PubMed
- number of protein molecules per cell (complex medium with amino acids, without glucose): 1656 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 10561 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 4162 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 3499 PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Mohamed Marahiel, Marburg University, Germany homepage
Your additional remarks
References
Martin Lehnik-Habrink, Marc Schaffer, Ulrike Mäder, Christine Diethmaier, Christina Herzberg, Jörg Stülke
RNA processing in Bacillus subtilis: identification of targets of the essential RNase Y.
Mol Microbiol: 2011, 81(6);1459-73
[PubMed:21815947]
[WorldCat.org]
[DOI]
(I p)
Onuma Chumsakul, Hiroki Takahashi, Taku Oshima, Takahiro Hishimoto, Shigehiko Kanaya, Naotake Ogasawara, Shu Ishikawa
Genome-wide binding profiles of the Bacillus subtilis transition state regulator AbrB and its homolog Abh reveals their interactive role in transcriptional regulation.
Nucleic Acids Res: 2011, 39(2);414-28
[PubMed:20817675]
[WorldCat.org]
[DOI]
(I p)
Noel Baichoo, Tao Wang, Rick Ye, John D Helmann
Global analysis of the Bacillus subtilis Fur regulon and the iron starvation stimulon.
Mol Microbiol: 2002, 45(6);1613-29
[PubMed:12354229]
[WorldCat.org]
[DOI]
(P p)
Jurgen J May, Nadine Kessler, Mohamed A Marahiel, Milton T Stubbs
Crystal structure of DhbE, an archetype for aryl acid activating domains of modular nonribosomal peptide synthetases.
Proc Natl Acad Sci U S A: 2002, 99(19);12120-5
[PubMed:12221282]
[WorldCat.org]
[DOI]
(P p)
Tamara Hoffmann, Alexandra Schütz, Margot Brosius, Andrea Völker, Uwe Völker, Erhard Bremer
High-salinity-induced iron limitation in Bacillus subtilis.
J Bacteriol: 2002, 184(3);718-27
[PubMed:11790741]
[WorldCat.org]
[DOI]
(P p)
J J May, T M Wendrich, M A Marahiel
The dhb operon of Bacillus subtilis encodes the biosynthetic template for the catecholic siderophore 2,3-dihydroxybenzoate-glycine-threonine trimeric ester bacillibactin.
J Biol Chem: 2001, 276(10);7209-17
[PubMed:11112781]
[WorldCat.org]
[DOI]
(P p)
B M Rowland, T H Grossman, M S Osburne, H W Taber
Sequence and genetic organization of a Bacillus subtilis operon encoding 2,3-dihydroxybenzoate biosynthetic enzymes.
Gene: 1996, 178(1-2);119-23
[PubMed:8921902]
[WorldCat.org]
[DOI]
(P p)
B M Rowland, H W Taber
Duplicate isochorismate synthase genes of Bacillus subtilis: regulation and involvement in the biosyntheses of menaquinone and 2,3-dihydroxybenzoate.
J Bacteriol: 1996, 178(3);854-61
[PubMed:8550523]
[WorldCat.org]
[DOI]
(P p)